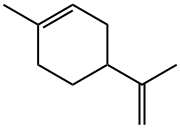
DL-Limonene
- CAS No.
- 138-86-3
- Chemical Name:
- DL-Limonene
- Synonyms
- D-LIMONENE;Dipentene;L-LIMONENE;Cinene;(-)-LIMONENE;1-methyl-4-(prop-1-en-2-yl)cyclohex-1-ene;LIMONENE(P);CITRUS TERPENE;DL-LIMONENE (MIXTURE OF D- AND L-FORM CA;FEMA 2633
- CBNumber:
- CB2178358
- Molecular Formula:
- C10H16
- Molecular Weight:
- 136.23
- MDL Number:
- MFCD00001558
- MOL File:
- 138-86-3.mol
| Melting point | -84--104 °C |
|---|---|
| Boiling point | 170-180 °C (lit.) |
| Density | 0.86 g/mL at 20 °C (lit.) |
| vapor density | 4.7 (vs air) |
| vapor pressure | <3 mm Hg ( 14.4 °C) |
| refractive index |
n |
| Flash point | 119 °F |
| storage temp. | Store below +30°C. |
| solubility | Chloroform: Slightly Soluble |
| form | Liquid |
| color | Clear colorless to pale yellow |
| Odor | Pleasant, pine-like; lemon-like. |
| Odor Threshold | 0.038ppm |
| explosive limit | 0.7-6.1%, 150°F |
| Odor Type | citrus |
| Water Solubility | <1 g/100mL |
| Merck | 14,5493 |
| BRN | 3587825 |
| Dielectric constant | 2.3(20℃) |
| Stability | Stable. Flammable. Incompatible with strong oxidizing agents. |
| InChIKey | AJSJXSBFZDIRIS-UHFFFAOYSA-N |
| LogP | 4.57 |
| Indirect Additives used in Food Contact Substances | DIPENTENE |
| FDA 21 CFR | 182.60 |
| CAS DataBase Reference | 138-86-3(CAS DataBase Reference) |
| EWG's Food Scores | 1-2 |
| FDA UNII | 9MC3I34447 |
| NCI Drug Dictionary | limonene, (+)- |
| NIST Chemistry Reference | Limonene(138-86-3) |
| EPA Substance Registry System | Limonene (138-86-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS07,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H226-H315-H317-H410 | |||||||||
| Precautionary statements | P210-P233-P240-P273-P280-P303+P361+P353 | |||||||||
| Hazard Codes | Xi,N | |||||||||
| Risk Statements | 10-38-43-50/53 | |||||||||
| Safety Statements | 24-37-60-61 | |||||||||
| RIDADR | UN 2052 3/PG 3 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | OS8350000 | |||||||||
| F | 8-10-23 | |||||||||
| Autoignition Temperature | 458 °F | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29021990 | |||||||||
| Toxicity | LD50 orally in Rabbit: 5300 mg/kg | |||||||||
| NFPA 704 |
|
DL-Limonene price More Price(33)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | W524905 | Dipentene mixtureofisomers | 138-86-3 | 1kg | $90.8 | 2024-03-01 | Buy |
| Sigma-Aldrich | CRM40448 | Limonene solution certified reference material, 2000?μg/mL in methanol, ampule of 1?mL | 138-86-3 | 1mL | $113 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.14546 | DL-Limonene (mixture of D- and L-form ~1:1) for synthesis | 138-86-3 | 1L | $78.1 | 2024-03-01 | Buy |
| Sigma-Aldrich | 334111 | Dipentene technical, for use as solvent (for the paint industry), mixture of various terpenes | 138-86-3 | 1l | $57.9 | 2023-06-20 | Buy |
| Sigma-Aldrich | 334111 | Dipentene technical, for use as solvent (for the paint industry), mixture of various terpenes | 138-86-3 | 4l | $144 | 2023-06-20 | Buy |
DL-Limonene Chemical Properties,Uses,Production
Description
D-limonene, which is a volatile oil, constitutes approximately 98% of orange peel oil by weight and has moderately good knockdown activity against ectoparasites of companion animals. The insecticidal activity of both d-limonene and linalool is enhanced when synergized by piperonyl butoxide. Apart from toxicoses reported in cats (65), d-limonene generally has a high margin of safety.
Description
(±)-Limonene is a cyclic monoterpene that has been found in various plant oils and Cannabis and has antifungal activity. It completely inhibits mycelial growth and aflatoxin B1 production in A. flavus when used at concentrations of 500 and 250 ppm, respectively. (±)-Limonene also inhibits the growth of various additional fungi in vitro, including S. cerevisiae, R. glutinis, and K. thermotolerans (MICs = 500-4,000 μg/ml). It has been identified as a contact dermatitis allergen in honing oil, paint thinner, and turpentine. Formulations containing (±)-limonene have been used as fragrance and flavoring ingredients.
Chemical Properties
Limonene (4R)-(+)-limonene and (4S)-(?)-limonene as well as the
racemate (dipentene) occur abundantly in many essential oils.The(+)
isomer is present in citrus peel oils at a concentration of over 90%; a low concentration
of the (?) isomer is found in oils from the Mentha species and conifers.
Limonene is a liquid with lemon-like odor. It is a reactive compound; oxidation
often yields more than one product. Dehydrogenation leads to p-cymene.
Limonene can be converted into cyclic terpene alcohols by hydrohalogenation,
followed by hydrolysis. Nitrosyl chloride adds selectively to the endocyclic double
bond; this reaction is utilized in the manufacture of (?)-carvone from (+)-
limonene.
(+)-Limonene is obtained in large amounts as a by-product in the production
of orange juice; (?)-limonene is isolated in relatively small quantities from essential
oils. Racemic limonenes, which are commercially available under the name
dipentene, are formed as by-products in many acid-catalyzed isomerizations of
α- and β-pinene. Distillation of the so-called dipentene fraction yields limonenes
in varying degrees of purity.
The limonenes are used as fragrance materials for perfuming household products
and as components of artificial essential oils.
Chemical Properties
d-, l- or dl-Limonene has a pleasant, lemon-like odor free from camphoraceous and turpentine-like notes. Limonene is the most important and widespread terpene; it is known in the d- and l- optically active forms and in the optically inactive dl-form (known as dipentene).
Chemical Properties
colourless or light yellow liquid
Occurrence
It has been reported found in more than 300 essential oils in amounts ranging from 90 to 95% (lemon, orange, mandarin) to as low as 1% (palmarosa); the most widespread form is the d-limonene, followed by the racemic form and then l-limo nene. Also reported found in ginger, nutmeg, pepper, mace, hop oil, coriander seed, calamus, dill herb, caraway seed and rosemary.
Toxicology
D-limonene is a clear colorless mobile liquid with a pleasant lemon-like odor. ((4R)-limonene is an optically active form of limonene having (4R)-configuration. It has a role as a plant metabolite. It is an enantiomer of a (4S)-limonene.)D-limonene is one of the most common terpenes in nature. It is a major constituent in several citrus oils (orange, lemon, mandarin, lime, and grapefruit). D-limonene is listed in the Code of Federal Regulations as generally recognized as safe (GRAS) for a flavoring agent and can be found in common food items such as fruit juices, soft drinks, baked goods, ice cream, and pudding.
Uses
Limonene is an antioxidant and flavoring agent that occurs in lemons, oranges, and pineapple juice, being obtained from the oils. It is a colorless liquid which is insoluble in water and propylene glycol, very slightly soluble in glycerin, and miscible with alcohol, most fixed oils, and mineral oil. It prevents or delays enzymatic browning-type oxidation.
Uses
d-Limonene is a flavoring agent that is a liquid, colorless with a pleasant odor resembling mild citrus. It is miscible in alcohol, most fixed oils, and mineral oil; soluble in glycerin; and insoluble in water and propylene glycol. It is obtained from citrus oil. It is also termed d-p-mentha-1,8,diene and cinene.
Definition
ChEBI: A monoterpene that is cyclohex-1-ene substituted by a methyl group at position 1 and a prop-1-en-2-yl group at position 4 respectively.
Preparation
d-Limonene may be obtained by steam distillation of citrus peels and pulp resulting from the production of juice and cold pressed oils, or from deterpenation of citrus oils; it is sometimes redistilled.
Production Methods
Limonene occurs in the oil of many plants and is the main constituent (≤86%) of the terpenoid fraction of fruit, flowers, leaves, bark, and pulp from shrubs, annuals, or trees including anise, mint, caraway, polystachya, pine, lime, and orange oil. It occurs as a by-product in the manufacture of terpineol and in various synthetic products made from α-pinene or turpentine oil. It is found in the gas phase of tobacco smoke and has been detected in urban atmospheres.
Aroma threshold values
Detection: 4 to 229 ppb
Taste threshold values
Taste characteristics at 30 ppm: sweet, orange, citrus and terpy.
Synthesis Reference(s)
Canadian Journal of Chemistry, 39, p. 1860, 1961 DOI: 10.1139/v61-249
Tetrahedron Letters, 20, p. 3519, 1979 DOI: 10.1016/S0040-4039(01)95450-4
General Description
A colorless liquid with an odor of lemon. Flash point 113°F. Density about 7.2 lb /gal and insoluble in water. Hence floats on water. Vapors heavier than air. Used as a solvent for rosin, waxes, rubber; as a dispersing agent for oils, resins, paints, lacquers, varnishes, and in floor waxes and furniture polishes.
Air & Water Reactions
Flammable. Insoluble in water.
Reactivity Profile
Cinene may react vigorously with strong oxidizing agents. May react exothermically with reducing agents to release hydrogen gas.
Health Hazard
Liquid irritates eyes; prolonged contact with skin causes irritation. Ingestion causes irritation of gastrointestinal tract.
Fire Hazard
Behavior in Fire: Containers may explode.
Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
Pharmaceutical Applications
Many studies have reported that D-limonene effectively plays a valuable role in the prevention of several chronic and degenerative diseases. This review provides worthy information about the beneficial effects of D-limonene such as antioxidant, antidiabetic, anticancer, anti-inflammatory, cardioprotective, gastroprotective, hepatoprotective, immune modulatory, anti-fibrotic, anti-genotoxic etc. This could in turn help in the application of D-limonene for clinical studies. Various plant families contain Terpenes as their secondary metabolites. Monoterpenes constitute an important part of these secondary metabolites. D-limonene is a well-identified monoterpene that is commonly applied as a fragrance ingredient in essential oils. D-limonene is known to possess remarkable biological activities. It can be effectively used for treating various ailments and diseases. Due to its diverse functions, it can be efficiently utilized for human health.
Numerous data on animal studies have highlighted the potential effects of D-limonene. D-limonene is well tolerated in experimental animals and protective effects of limonene were observed in several preclinical models. Animal studies showing the cardio protective and hepatoprotective effects of D-limonene are found to be inadequate. Moreover, studies exploring the anticancer efficacy of D-limonene on animal models are also found to be limited. The study on the effect of D-limonene on humans is also extremely limited. Therefore, our present review will open new avenues to undertake further research on D-limonene in preclinical models and for subjecting this compound to effective clinical trials.
Contact allergens
Limonene is a racemic form of dand l-limonene. d-Limonene is contained in Citrus species such as citrus, orange, mandarin, and bergamot. l-Limonene is contained in Pinus pinea. The racemic form (dand l-limonene) is also named dipentene.
Contact allergens
Dipentene corresponds to a racemic mixture of d-limonene and l-limonene. Dipentene can be prepared from wood turpentine or by synthesis. It is used as a solvent for waxes, rosin and gums, in printing inks, perfumes, rubber compounds, paints, enamels, and lacquers. An irritant and sensitizer, dipentene caused contact dermatitis mainly in painters, polishers, and varnishers
Anticancer Research
Tested as promising antitumor molecules in induced tumor on rat tissues, D-limonenewas tested in preclinical studies in patients with advanced cancer. Limonene inhibitsthe activity of HMG-CoA reductase, subsequently reducing the possibility of cancergrowth. The mechanism of action involves the inhibition of prenyltransferases withthe activation of glutathione-S transferase and uridine diphospho-glucuronosyltransferase.More interest was pointed on the principal metabolite:perillyl alcohol which is more potent than limonene. The interest on perillyl alcoholis based on the necessity of a very high dosage of D-limonene in preclinical studies(about 1000 mg/kg/day in human mammary tumor) that can cause notably importantside effects. The more active perillyl alcohol and the less low active doseshypothesized this molecule as a clinical candidate (Pattanayak et al. 2009; Chenet al. 2013; Fontes et al. 2013; Rani and Sharma 2013).
Safety Profile
A skin irritant. Flammable when exposed to heat or flame; can react vigorously with oxidzing materials. When heated to decomposition it emits acrid smoke and irritating fumes.
Toxicology
D-limonene is considered to have fairly low toxicity. It has been tested for carcinogenicity in mice and rats. Although initial results showed d-limonene increased the incidence of renal tubular tumors in male rats, female rats and mice in both genders showed no evidence of any tumor. Subsequent studies have determined how these tumors occur and established that d-limonene does not pose a mutagenic, carcinogenic, or nephrotoxic risk to humans. In humans, d-limonene has demonstrated low toxicity after single and repeated dosing for up to one year. Being a solvent of cholesterol, d-limonene has been used clinically to dissolve cholesterol-containing gallstones. Because of its gastric acid neutralizing effect and its support of normal peristalsis, it has also been used for relief of heartburn and gastroesophageal reflux (GERD). D-limonene has well-established chemopreventive activity against many types of cancer. Evidence from a phase I clinical trial demonstrated a partial response in a patient with breast cancer and stable disease for more than six months in three patients with colorectal cancer.
Potential Exposure
Dipentene is used as a solvent; in rubber compounding and reclamation; and to make paints,enamels, lacquer, and perfumes.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit
Carcinogenicity
Induction of kidney neoplasias has been observed in male rats of strains that have significant concentrations of the protein a2u-globulin (158a). This protein is not expressed in females or species other than the rat; therefore, limonene carcinogenicity appears to be limited to the male of specific strains of this species. Subcutaneous injection of the compound or its hydroperoxide into C57BL/6 mice decreased the incidence of dibenzopyrene- induced tumors appreciably. Given orally either 15 min or 1 h prior to nitrosodiethylamine, D-limonene reduced forestomach tumor formation by about 60% and pulmonary adenoma formation by about 35%. Reduction of cancer incidence and metastasis by limonene has also been reported in other systems (158b).
Environmental Fate
Limonene is insoluble and is stable in water. Substances like limonene that are monoterpenes are released in large amounts mainly to the atmosphere. The chemical and physical properties of limonene also indicate that limonene is distributed mainly to air. Based on the physical and chemical properties of limonene, when this substance is released to ground, it has low to very low mobility in soil. The soil adsorption coefficient (Koc), calculated on the basis of the solubility (13.8 mg l-1 at 25 ℃) and the log octanol/water partition coefficient (4.232), ranges from 1030 to 4780.3. Henry’s Law constant indicates that limonene rapidly volatilizes from both dry and moist soil; however, its strong adsorption to soil may slow this process. In the aquatic environment, limonene is expected to adsorb to sediment and suspended organic particles to rapidly volatilize to the atmosphere, based on its physical and chemical properties. The estimated half-life for volatilization of limonene from a model river (1 m deep, flow 1 ms-1, and wind speed 3 ms-1) is 3.4 h. The bioconcentration factor, calculated on the basis of water solubility and the log octanol/water partition coefficient, is 246–262, suggesting that limonene may accumulate in fish and other aquatic organisms.
storage
Color Code—Red: Flammability Hazard: Store ina flammable liquid storage area or approved cabinet awayfrom ignition sources and corrosive and reactive materials.Store in tightly closed containers in a cool, well-ventilatedarea. Before entering confined space where this chemicalmay be present, check to make sure that an explosive concentration does not exist. Store in tightly closed containersin a cool, well-ventilated area away from heat and incompatible materials. Sources of ignition, such as smoking andopen flames, are prohibited where this chemical is used,handled, or stored in a manner that could create a potentialfire or explosion hazard. Metal containers involving thetransfer of 5 gallons or more of this chemical should begrounded and bonded. Drums must be equipped with selfclosing valves, pressure vacuum bungs, and flame arresters.Use only nonsparking tools and equipment, especially whenopening and closing containers of this chemical.
Shipping
This compound requires a shipping label of“FLAMMABLE LIQUID.” It falls in Hazard Class 3 andPacking Group II.
Toxicity evaluation
D-Limonene can cause a renal syndrome in male rats. Male rats
have a background of spontaneous protein droplets in the
proximal tubule, particularly within the cells of the P2 segment.
D-Limonene can increase the formation of protein droplets,
and experiments show that the only protein accumulating is
α2u-globulin. α2u-Globulin is synthesized in the liver, then it is
released into the general circulation and reabsorbed by renal
proximal tubule cells. Synthesis of α2u-globulin occurs only in
adult male rats and to understand the critical role of α2uglobulin
in the renal effects of D-limonene the absence of
histopathological changes can be studied in female rats, since
they cannot produce α2u-globulin. It was concluded from these
studies that D-limonene does not have a renal toxicity effect on
species that do not synthesize α2u-globulin, for example,
female rats, male and female mice, and dogs.
To develop nephropathy, there is a prerequisite step in
which an agent binds to α2u-globulin and in the case of
D-limonene, this agent is 1,2-epoxide. Binding of this agent to
α2u-globulin reduces the rate of degradation relative to that of
native protein, thereby causing it to accumulate. Accumulation
of α2u-globulin can be observed after a single oral dose of
D-limonene and continued treatment results in additional
histological changes in the kidney. Renal function can be
observed by increasing the amount of α2u-globulin in urine.
These functional changes occur only in male rats and only at
doses that exacerbate protein droplet formation. In response to
the cell death and functional changes, there is an increase in cell
proliferation in the kidney. With continued treatment, cell
proliferation persists but it does not restore renal function.
Increasing cell proliferation is directly related to the development
of renal tubular tumors and is dependent on the presence
of α2u-globulin. α2u-Globulin nephropathy and renal cell
proliferation occur at the same doses consistent with those that
produce renal tubular tumors.
α2u-Globulin is one type of superfamily proteins that bind
and transport a variety of agents. Many of these proteins are
synthesized in mammalian species, including humans. The
protein that can be found in human urine is very different from
that in rat urine. About 1% of the protein concentration in male
rat urine can be found in human urine. Human urinary protein
is predominantly a species of high molecular mass, and there is
no protein in human plasma or urine identical to α2u-globulin
and there is no protein-like α2u-globulin detected in human
kidney tissue. Even though the binding of D-limonene-
1,2-epoxide to α2u-globulin can be shown in vitro, other
superfamily proteins, especially those synthesized by humans,
do not bind 1,2-epoxide.
There is no verification that any human protein can
contribute to a renal syndrome similar to α2u-globulin
nephropathy, and consequently no evidence can be found that
D-limonene is carcinogenic in humans by a mechanism similar
to α2u-globulin nephropathy. The initiation of renal cell
tumors in male rats by binding to agents that act through an
α2u-globulin-associated response is not projected as a carcinogenic
hazard to humans. This can be concluded based on an
extensive evidence that the presence of α2u-globulin is an
absolute requirement for the carcinogenic activity. Therefore,
D-limonene has no carcinogenic activity in human since neither
α2u-globulin nor any protein that can function like α2uglobulin
is synthesized by humans.
DL-Limonene Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of2
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| ShanDong Look Chemical Co.,Ltd. | +8617653113219 | sales01@sdlookchemical.com | China | 2739 | 58 |
| Shandong Zhishang New Material Co., Ltd. | +8617653113209 | sales002@sdzschem.com | China | 3050 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +86-89586680 +86-13289823923 | 1026@dideu.com | China | 9020 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | sales@hbmojin.com | China | 12471 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 | peter@yan-xi.com | China | 6000 | 58 |
| Hebei Kingfiner Technology Development Co.Ltd | +86-15532196582 +86-15373005021 | lisa@kingfinertech.com | China | 3001 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 18232 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | info@dakenam.com | China | 15957 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21689 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | info@tnjchem.com | China | 2989 | 55 |
Related articles
- DL-Limonene: Reactions, biosynthesis and applications
- D-limonene is a clear colorless mobile liquid with a pleasant lemon-like odor. It is the major component in the oil of citrus ....
- May 19,2023
- Introduction of D-limonene
- D-limonene is a clear colorless mobile liquid with a pleasant lemon-like odor.
- Jan 6,2022
View Lastest Price from DL-Limonene manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2023-12-26 | DL-Limonene
138-86-3
|
US $100.00-1.00 / KG | 1KG | 99% | g-kg-tons, free sample is available | Henan Fengda Chemical Co., Ltd | |
 |
2023-10-30 | DL-Limonene
138-86-3
|
US $0.00-0.00 / kg | 1kg | 99% | 50000kg | Hebei Kingfiner Technology Development Co.Ltd | |
 |
2023-09-04 | Dipentene
138-86-3
|
US $20.00 / KG | 1KG | 0.99 | 20MT/MON | Hebei Yanxi Chemical Co., Ltd. |
-

- DL-Limonene
138-86-3
- US $100.00-1.00 / KG
- 99%
- Henan Fengda Chemical Co., Ltd
-

- DL-Limonene
138-86-3
- US $0.00-0.00 / kg
- 99%
- Hebei Kingfiner Technology Development Co.Ltd
-

- Dipentene
138-86-3
- US $20.00 / KG
- 0.99
- Hebei Yanxi Chemical Co., Ltd.