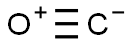CARBON MONOXIDE suppliers
CARBON MONOXIDE
- CAS:
- 630-08-0
- MF:
- CO
- MW:
- 28.01
Suppliers by country/region
Company Type
Properties
- Melting point:
- −205 °C(lit.)
- Boiling point:
- −191.5 °C(lit.)
- Density
- d4-195 (liq) 0.814; d (gas) 0.968 (air = 1.000); d40 at 760 mm: 1.250 g/liter
- vapor density
- 0.97 (vs air)
- vapor pressure
- >760 mmHg at 20 °C
- refractive index
- 1.0003
- solubility
- At 20 °C and at a pressure of 101 kPa, 2.266 volumes of carbon monoxide dissolve in 100 volumes of water.
- form
- colorless gas
- color
- colorless
- Odor
- Odorless gas
- explosive limit
- 74%
- Water Solubility
- mL/100mL H2O: 3.3 (0°C), 2.3 (20°C) [MER06]
- Merck
- 13,1823
- BRN
- 3587264
- Exposure limits
- TLV-TWA 50 ppm (~55 mg/m3) (ACGIH, MSHA, and OSHA); STEL 400 ppm (ACGIH); IDLH 1500 ppm (NIOSH).
- InChIKey
- UGFAIRIUMAVXCW-UHFFFAOYSA-N
Safety Information
- Symbol(GHS)




GHS02,GHS04,GHS06,GHS08
- Signal word
- Danger
- Hazard statements
- H220-H280-H331-H360D-H372
- Precautionary statements
- P202-P210-P304+P340+P311-P308+P313-P403+P233-P410+P403
- Hazard Codes
- F+,T
- Risk Statements
- 61-12-23-48/23
- Safety Statements
- 53-45
- RIDADR
- UN 1016 2.3
- WGK Germany
- 1
- RTECS
- FG3500000
- Autoignition Temperature
- 609 °C
- DOT Classification
- 2.3, Hazard Zone D (Gas poisonous by inhalation)
- HazardClass
- 2.3
- Toxicity
- LC50 inhal (rat) 1807 ppm (2065 mg/m3; 4 h)
LCLO inhal (man) 4000 ppm (4570 mg/m3; 30 min)
PEL (OSHA) 50 ppm (55 mg/m3)
TLV-TWA (ACGIH) 25 ppm (29 mg/m3)
- IDLA
- 1,200 ppm
Use
Carbon monoxide bonds to the iron in hemoglobin to form carboxyhemoglobin, which interferes with oxygen’s ability to bind to hemoglobin to form oxyhemoglobin. Thus carbon monoxide is a chemical asphyxiant, which prevents oxygen from reaching body tissues.Carbon monoxide is an important industrial chemical. It is produced, along with hydrogen, by steam reforming. Carbon monoxide is a basic feedstock in these processes. Carbon monoxide is also useful as a reducing agent. It is used in metallurgy to obtain metals from their oxides. For example, during iron and steel production coke in a blast furnace is converted to carbon monoxide. The carbon monoxide reduces the Fe3+ in the iron (III) oxide contained in the iron ore to produce elemental iron according to the reaction: Fe2O3(s) + 3CO(g) → 2Fe(l) + 3CO2(g).
76 supplier list of "CARBON MONOXIDE"
- Product Name:carbon monoxide,cyl. with 10 L (net~3kg)
- Company Type: Reagent
- Country/Region: CHINA
- Main Products: Gamma-butyrolactone

- Product Name:Carbon monoxide
- Products Intro:Purity: 99% | Package: 1 kg,5 kg, 10 kg,25kg and 1 MT
- Company Type: Manufacturer
- Country/Region: INDIA

- Product Name:Carbon monoxide
- Products Intro:Purity: 98% | Package: 1 kg,5 kg, 10 kg,25kg
- Company Type: Manufacturer
- Country/Region: INDIA

- Product Name:Carbon monoxide
- Products Intro:Purity: 98% | Package: 1 kg,5 kg, 10 kg,25kg and 1 MT
- Company Type: Reagent
- Country/Region: INDIA

- Product Name:Carbonmonoxide
- Products Intro:Brand:sigma aldrich | Product Number:295116 | Purity:≥99.0%
- Company Type: Trader
- Country/Region: INDIA