| | 3333-67-3 Basic information More.. |
| Product Name: | Nickel carbonate | | Synonyms: | nickelmonocarbonate;PHENOLSTANDARD,SOLUTION;Nickel Carbonate, Powder;Carbonic acid nickel(II) salt;Nickel (II) carbonate≥98%;c.i.77779;carbonicacid,nickel(2+)salt;Carbonicacid,nickel(2+)salt(1:1) | | CAS: | 3333-67-3 | | MF: | CNiO3 | | MW: | 118.7 | | EINECS: | 222-068-2 | | Mol File: | 3333-67-3.mol | 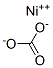 |
Use
Nickel carbonate is the starting material for preparing many nickel salts. It reacts with dilute acids evolving carbon dioxide, and upon evaporation of the solution corresponding nickel salts are formed. The nitrate, sulfate and phosphate salts are prepared from carbonate. Similarly, reactions with hydrofluoric, hydrochloric, hydrobromic, or hydriodic acids yield hydrated nickel halides: namely NiF24H2O, NiCl26H2O, NiBr26H2O, and NiI26H2O, respectively:
NiCO3 + HCl NiCl26H2O + CO2
NICKEL CARBONATE 611Nickel carbonate decomposes to nickel oxide when strongly ignited:
NiCO3 NiO + CO2
Nickel carbonate, when dissolved in aqueous thiocyanic acid, yields a yellow brown precipitate of hydrated nickel thiocyanate:
2 NiCO3 + 2HSCN Ni(SCN)2 + CO2 + H2O
Nickel carbonate forms many double salts, such as, Na2CO3NiCO310H2O with alkali metal carbonates. However, such double carbonates usually are prepared by mixing an alkali metal or ammonium bicarbonate solution with a nickel salt solution, followed by crystallization.
- Nickel Carbonate
-

- US $120.00 / kg
- 2024-05-21
- CAS:3333-67-3
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10ton
- Nickel carbonate
-

- US $6.00 / kg
- 2024-05-20
- CAS:3333-67-3
- Min. Order: 1kg
- Purity: More than 99%
- Supply Ability: 2000KG/Month
- Nickel carbonate
-

- US $6.00 / KG
- 2024-05-01
- CAS:3333-67-3
- Min. Order: 1KG
- Purity: More than 99%
- Supply Ability: 2000KG/Month
|
|
3333-67-3
Recommend Suppliers |
|