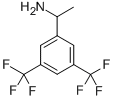
(RS)-1-[3,5-BIS(TRIFLUOROMETHYL)PHENYL]ETHYLAMINE synthesis
- Product Name:(RS)-1-[3,5-BIS(TRIFLUOROMETHYL)PHENYL]ETHYLAMINE
- CAS Number:187085-97-8
- Molecular formula:C10H9F6N
- Molecular Weight:257.18
![1-[3,5-Bis(trifluoroMethyl)phenyl]ethanaMine Hydrochloride](/CAS/20150408/GIF/374822-27-2.gif)
374822-27-2
31 suppliers
$45.00/50mg
![(RS)-1-[3,5-BIS(TRIFLUOROMETHYL)PHENYL]ETHYLAMINE](/CAS/GIF/187085-97-8.gif)
187085-97-8
24 suppliers
$65.00/250mg
Yield:187085-97-8 100%
Reaction Conditions:
with sodium hydroxide in water;
Steps:
5 DESCRIPTION 5; (RS)-α-Methyl-3,5-bis(trifluoromethyl)benzenemethanamine
O-Methylhydroxylamine hydrochloride (19.57 g, 234 mmol) was added to a solution of 1-[3,5-bis(trifluoromethyl)phenyl]ethanone (50 g, 195 mmol), and sodium acetate (31.9 g, 234 mmol) in ethanol (450 mL) and water (150 mL) and the mixture was heated under reflux for 18 hours. The mixture was cooled, water (1000 mL) was added and the mixture was extracted with diethyl ether. The combined organic fractions were dried (MgSO4) and the solvent was evaporated under reduced pressure. The residue was dissolved in tetrahydrofuran (100 mL), cooled to 020 C. and borane-tetrahydrofuran complex (1M in tetrahydrofuran, 730 mL) was added. The mixture was heated under reflux for 18 hours, cooled and hydrochloric acid (5M, 500 mL) was added slowly. The tetrahydrofuran was evaporated under reduced pressure and the mixture was basified with aqueous sodium hydroxide (4M). The mixture was extracted with ether and the combined organic fractions were washed with aqueous sodium hydroxide (4M) and dried (MgSO4). and ethereal hydrogen chloride (400 mL, 400 mmol) was added. The solid was collected, washing with diethyl ether, and dried in vacuo to give (RS)-α-methyl-3,5-bis(trifluoromethyl)benzenemethanamine hydrochloride as a colorless solid (48.2 g, 100%). 1H NMR (400 MHz, CD3OD) δ8.13 (2H, s), 8.07 (1H, s), 4.73 (1H, q, J 6.8 Hz), 3.31 (2H, d, J 1.4 Hz), and 1.69 (3H, d, J 6.8 Hz). m/z (ES+) 258 (M+1). A sample was suspended in aqueous sodium hydroxide (4M) and extracted with ether. The combined organic fractions were washed with aqueous sodium hydroxide (4M), dried (MgSO4) and the solvent was evaporated under reduced pressure to give the title compound as a colorless oil. 1H NMR (400 MHz, CDCl3) δ7.85 (2H, s), 7.76 (1H, s), 4.30 (1H, q, J 6.6 Hz), 1.74 (2H, br s), and 1.42 (3H, d, J 6.6 Hz). m/z (ES+) 258 (M+1).
References:
US2003/225059,2003,A1 Location in patent:Page 21
![1H-Isoindole-1,3(2H)-dione, 2-[1-[3,5-bis(trifluoromethyl)phenyl]ethyl]-](/CAS/20210305/GIF/690653-26-0.gif)
690653-26-0
0 suppliers
inquiry
![(RS)-1-[3,5-BIS(TRIFLUOROMETHYL)PHENYL]ETHYLAMINE](/CAS/GIF/187085-97-8.gif)
187085-97-8
24 suppliers
$65.00/250mg