| Identification | Back Directory | [Name]
F 12158 | [CAS]
194468-36-5 | [Synonyms]
Javlor
F 12158
BMS 710485
Ditartrate
Vinflunine Ditartrate
20',20'-Difluoro-3',4'-dihydrovinorelbine Ditartrate
4'-Deoxy-20',20'-difluoro-5'-norvincaleukoblastine Ditartrate
(2β,3β,4β,5α,12β,19α)-4-(Acetyloxy)-6,7-didehydro-15-[(2R,4R,6S,8S)-4-(1,1-difluoroethyl)-1,3,4,5,6,7,8,9-octahydro-8-(Methoxycarbonyl)-2,6-Methano-2H-azecino[4,3-b]indol-8-yl]-3-hydroxy-16-Methoxy-1-MethylaspidosperMidine-3-carboxylic Acid Methyl Ester (2R,3R)-2,3-Dihydroxybutanedioate | [Molecular Formula]
C49H59F2N4O14 | [MDL Number]
MFCD12756258 | [MOL File]
194468-36-5.mol | [Molecular Weight]
966.008 |
| Chemical Properties | Back Directory | [Appearance]
White Solid | [Melting point ]
244-246°C (dec) | [storage temp. ]
-20°C Freezer | [solubility ]
DMSO (Slightly), Methanol (Slightly) | [form ]
Solid | [color ]
White to Light Beige |
| Hazard Information | Back Directory | [Chemical Properties]
White Solid | [Uses]
Semisynthetic Vinca alkaloid with microtubule destabilizing and antiangiogenic activity; derivative of Vinorelbine. Antineoplastic | [Uses]
Vinflunine is a semisynthetic Vinca alkaloid with microtubule destabilizing and antiangiogenic activity. Vinflunine is a derivative of Vinorelbine. Vinflunine is used as an antineoplastic. | [Clinical Use]
Vinflunine ditartrate is a second generation difluorinated analog of the naturally-occuring substance
vinorelbine and it is approved for the treatment of non-small cell lung cancer, metastatic breast cancer
and ovarian cancer. Vinflunine, a tubulin polymerization inhibitor, belongs to the vinca alkaloid class
of anti-cancer agents. Introduction of the difluoro group of vinflunine dramatically improved antitumor
activity of the parent vinorelbine structure. Vinflunine was discovered by Pierre Fabre
Laboratories and in 2004 was licensed to Bristol-Myers Squibb for development and commercialization.
In 2007, the rights to venflunine were returned to Pierre Fabre which completed its development. | [Synthesis]
Vinflunine can be prepared directly from vinorelbine (155) through the use of superacid chemistry. Reaction of 155 with antimony pentaflouride in hydrofluoric acid and Nbromosuccinimide
followed by treatment with two equivalents of tartaric acid produced vinflunine
ditartrate (XV) in 25% yield. An alternative synthesis of vinflunine was realized through reaction
of vinblastine or 3?ˉ,4?ˉ-dihydrovinblastine (156) with antimony pentaflouride and hydrofluoric acid in
chloroform to give the difluoro alkaloid 157 in 40% yield Ring contraction was effected by
reaction with trifluoroacetic acid and N-bromosuccinimide followed by aqueous sodium bicarbonate and
silver tetrafluoroborate to give vinflunine in 88% yield. Vinflunine ditartrate (XV) was prepared by
treating a solution of vinflunine in toluene with two equivalents of tartaric acid.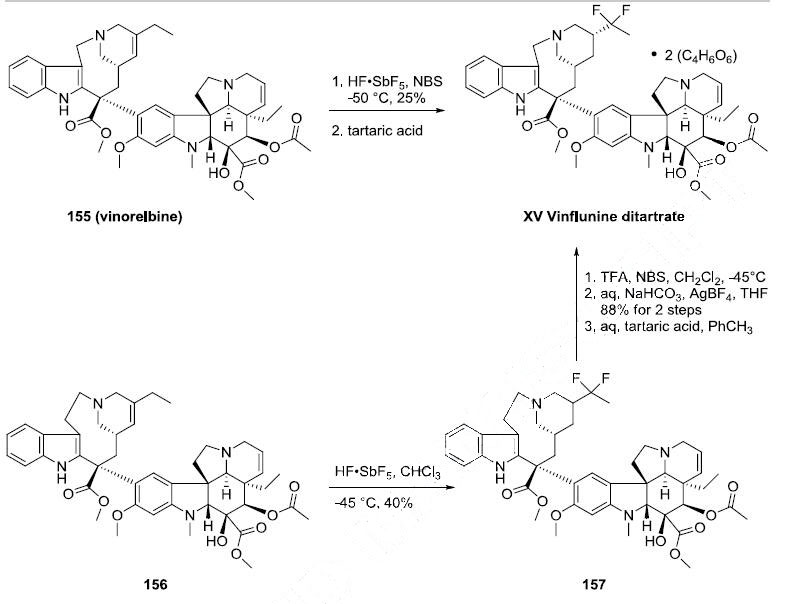
|
|
| Company Name: |
Energy Chemical
|
| Tel: |
021-58432009 400-005-6266 |
| Website: |
http://www.energy-chemical.com |
| Company Name: |
InvivoChem
|
| Tel: |
13549236410 |
| Website: |
https://www.invivochem.cn/ |
|