130636-61-2
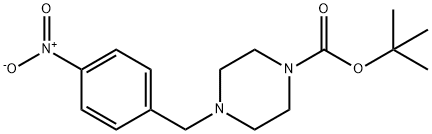 130636-61-2 结构式
130636-61-2 结构式
基本信息
1-Boc-4-(4-nitrobenzyl)piperazine
1-[(4-nitrophenyl)methyl]-4-tert-butyloxycarbonylpiperazine
tert-butyl 4-[(4-nitrophenyl)Methyl]piperazine-1-carboxylate
4-(4-NITROBENZYL)PIPERAZINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER
4-(4-NITROBENZYL)PIPERAZINE-1-CARBOXYLIC ACID TERT-BUTYL ESTER, 95+%
4-[(4-nitrophenyl)methyl]-1-piperazinecarboxylic acid tert-butyl ester
4-[(4-NITROPHENYL)METHYL]-1-PIPERAZINECARBOXYLIC ACID, 1,1-DIMETHYLETHYL ESTER
1-Piperazinecarboxylic acid, 4-[(4-nitrophenyl)methyl]-, 1,1-dimethylethyl ester
物理化学性质
制备方法

100-11-8

57260-71-6

130636-61-2
以4-硝基溴化苄和N-BOC-哌嗪为原料合成4-(4-硝基苯)哌嗪-1-羧酸叔丁酯的一般步骤:向含有1-哌嗪羧酸叔丁酯(0.74g,4mmol)和4-硝基苄基溴(0.86g,4mmol)的N,N-二甲基甲酰胺(DMF,5mL)溶液中加入碳酸钾(3g)。反应混合物在室温下搅拌4小时。反应完成后,用乙酸乙酯(50mL)萃取产物,有机相用水(20mL×3)洗涤。有机层用无水硫酸钠干燥,随后在减压下浓缩去除溶剂。粗产物通过柱色谱法纯化,以乙酸乙酯为洗脱剂,得到目标化合物4-(4-硝基苄基)-1-哌嗪甲酸叔丁酯(3)(1.22g,收率95%),熔点为99-100℃。1H NMR(DMSO-d6)数据如下:δ 8.21-8.19(d,2H,J = 8.8Hz,Ar-H),7.61-7.59(d,2H,J = 8.0Hz,Ar-H),3.63(s,2H,CH2),3.34-3.30(m,4H,部分模糊,2×CH2),2.35-2.32(m,4H,2×CH2),1.39(s,9H,3×CH3)。元素分析(C16H23N3O4)结果符合计算值。
参考文献:
[1] Bioorganic and Medicinal Chemistry, 2005, vol. 13, # 19, p. 5592 - 5599
[2] Patent: US2004/34033, 2004, A1. Location in patent: Page/Page column 12; Sheet 1
[3] Patent: US2007/249649, 2007, A1. Location in patent: Page/Page column 116
[4] Patent: WO2012/110773, 2012, A1. Location in patent: Page/Page column 120
[5] Patent: WO2014/27199, 2014, A1. Location in patent: Page/Page column 122
