151907-80-1
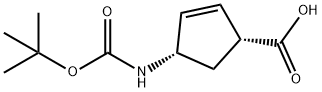 151907-80-1 结构式
151907-80-1 结构式
基本信息
(1R,4S)-4-(叔丁氧羰基氨基)环戊-2-烯甲酸
(1R,4S)- N-BOC-4-氨基环戊-2-烯甲酸
(1R,4S)-4-((叔丁氧羰基)氨基)环戊-2-烯羧酸
(1R,4S)-(+)-4-(BOC-氨基)-2-环戊烯羧酸
(+)-(1R,4S)-N-BOC-4-氨基环戊-2-烯羧酸
(+)-(1R,4S)- N-BOC-4-氨基环戊-2-烯甲酸
(+)-(1R,4S)-N-BOC-GAMMA-HOMOCYCLOLEU-2-ENE
(1R,4S)-Boc-4-aminocyclopent-2-ene-carboxylic acid
(1R,4S)-4-Boc-aMino-cyclopent-2-enecarboxylic acid
(1S,4R)-N-BOC-1-AMINOCYCLOPENT-2-ENE-4-CARBOXYLIC ACID
(+)-(1R,4S)-N-BOC-4-AMINOCYCLOPENT-2-ENECARBOXYLIC ACID
Cis-(1R,4S)-4-Boc-aMino-2-Cyclopentene-1-carboxylic acid
(+)-(1R,4S)-N-BOC-4-AMINOCYCLOPENT-2-ENE-4-CARBOXYLIC ACID
(+)-(1S,4R)-N-BOC-1-AMINOCYCLOPENT-2-ENE-4-CARBOXYLIC ACID
(1R,4S)-(+)-4-(BOC-AMINO)-2-CYCLOPENTENE-1-CARBOXYLIC ACID
物理化学性质
制备方法
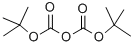
24424-99-5
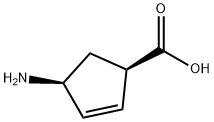
134003-04-6

151907-80-1
将(1R,4S)-4-氨基环戊-2-烯-1-羧酸(130g,1.0mol)溶于水(250mL)和THF(750mL)的混合溶剂中,加入碳酸氢钠(170g,2.0mol)。搅拌30分钟后,缓慢加入固体二碳酸二叔丁酯(230g,1.05mol)。反应混合物在室温下搅拌过夜。反应完成后,过滤除去不溶物,蒸发去除THF。将残余物冷却至0℃,缓慢加入2N HCl水溶液(约500mL)调节pH至3。析出的白色固体经过滤收集,用水洗涤,真空干燥过夜,得到(1R,4S)-4-((叔丁氧羰基)氨基)环戊-2-烯甲酸(230g,100%)。产物通过1H NMR(400MHz,CD3OD)表征:δ 5.95(m,1H),5.79(m,1H),4.80(br s,1H),3.45(m,1H),2.50(m,1H),1.79(m,1H),1.44(s,9H)。
参考文献:
[1] Patent: WO2004/76411, 2004, A2. Location in patent: Page 27-28
[2] Patent: WO2003/93231, 2003, A2. Location in patent: Page/Page column 55-56
[3] Patent: US2007/155731, 2007, A1. Location in patent: Page/Page column 27
[4] Patent: US2007/155731, 2007, A1. Location in patent: Page/Page column 35
[5] Patent: WO2005/110409, 2005, A2. Location in patent: Page/Page column 30
| 报价日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
| 2026/03/03 | 36182 | (1S,4R)-N-BOC-1-氨基环戊-2-烯-4-甲酸 (1S,4R)-N-BOC-1-Aminocyclopent-2-ene-4-carboxylic acid, 95%, 98% ee, Thermo Scientific™ | 151907-80-1 | 1g | 4277元 |
| 2025/05/22 | XW0215190780104 | (1R,4S)-N-BOC-4-氨基环戊-2-烯甲酸 | 151907-80-1 | 10G | 993元 |
| 2025/05/22 | XW0215190780103 | (1R,4S)-N-BOC-4-氨基环戊-2-烯甲酸 | 151907-80-1 | 5G | 646元 |
