338990-31-1
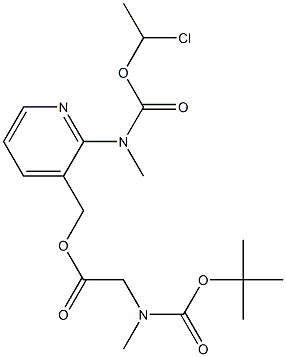 338990-31-1 结构式
338990-31-1 结构式
基本信息
艾莎康唑侧链
艾沙康唑侧链中间体
艾沙康唑侧链中间体1
艾沙康唑侧链中间体C2
艾沙康唑侧链中间体C1
艾沙康唑侧链中间体 10G
侧链中间体 CAS:338990-31-1
N-甲基-N-(3-[((N-叔丁氧羰基-N-甲基氨基)乙酰氧基)甲基]吡啶-2-基)氨基甲酸(1-
N-甲基-N-(3-[((N-叔丁氧羰基-N-甲基氨基)乙酰氧基)甲基]吡啶-2-基)氨基甲酸(1-氯乙基)酯
(2-(((1-chloroethoxy)carbonyl)(methyl)amino)pyridin-3-yl)methyl N-(tert-butoxycarbonyl)-N-methylglycinate
[N-methyl-N-3-((tert-butoxycarbonylmethylamino)acetoxymethyl)pyridin-2-yl]carbamic acid 1-chloroethyl ester
(2-(((1-chloroethoxy)carbonyl)(Methyl)aMino)pyridin-3-yl)Methyl 2-((tert-butoxycarbonyl)(Methyl)aMino)acetate
N-[(1,1-diMethylethoxy)carbonyl]-N-Methyl-, [2-[[(1-chloroethoxy)carbonyl]MethylaMino]-3-pyridinyl]Methyl ester
N-Methyl-N-(3-[((N-tert-butoxycarbonyl-N-methylamino)acetoxy)methyl]pyridin-2-yl)carbamic acid 1-chloroethyl ester
1-diMethylethoxy)carbonyl]-N-Methyl [2-[[(1-chloroethoxy)carbonyl]MethylaMino]-3-pyridinyl]Methyl esterChemicalBook
Glycine, N-[(1,1-diMethylethoxy)carbonyl]-N-Methyl-, [2-[[(1-chloroethoxy)carbonyl]MethylaMino]-3-pyridinyl]Methyl ester
Glycine, N-[(1,1-diMethylethoxy)carbonyl]-N-Methyl-, [2-[[(1-chloroethoxy)carbonyl]MethylaMino]-3-pyridinyl]Methyl esterChemicalBook
N-methyl-N- (3-[((N-tert-butoxycarbonyl-N-methylamino) acetoxy) methyl] pyridin-2-yl) carbamic acid (1-chloroethyl) ester moxa Shaconazole side chain
物理化学性质
制备方法

50893-53-3

13734-36-6

32399-12-5

338990-31-1
h)N-甲基-N-(3-[((N-叔丁氧羰基-N-甲基氨基)乙酰氧基)甲基]吡啶-2-基)氨基甲酸(1-氯乙基)酯的制备:将2-甲胺基-3-吡啶甲醇(22g,0.159mol)和二异丙胺(36.1mL,0.207mol,1.3当量)溶解于二氯甲烷(1L)中,并在乙醇-冰浴中冷却至约-13℃。在1小时内缓慢滴加1-氯乙基氯甲酸酯(17.5mL,0.161mol,1.01当量),滴加完毕后继续搅拌反应混合物1小时。随后加入2-((叔丁氧羰基)(甲基)氨基)乙酸(39.2g,0.207mol,1.3当量)和WSC(39.7g,0.207mol,1.3当量),分批次在10分钟内加入。加入DMAP(5.8g,0.047mol,0.3当量)后,将反应混合物于-7℃下搅拌2小时。反应完成后,将混合物升温至25℃并浓缩,残余物溶解于二乙醚(1L)中。将醚溶液转移至分液漏斗,依次用0.1N HCl(500mL×3)、水(500mL)、饱和NaHCO3水溶液(500mL)和饱和盐水(500mL×2)洗涤。有机相经无水MgSO4干燥后,减压浓缩得到粗产物(48.2g,收率约72.9%),无需进一步纯化即可用于后续反应。1H-NMR(270MHz,CDCl3):δ1.42(9H,d,J=24.1Hz),1.57(1.5H,br.s),1.88(1.5H,br.s),2.94(3H,s),3.37(3H,s),4.00(2H,d,J=21.1Hz),5.18(2H,d,J=13.5Hz),6.58(1H,q,J=5.45,11.0Hz),7.30(1H,s),7.82(1H,d,J=6.9Hz),8.47(1H,s);FAB-MS:m/z 416(M+H)+。
参考文献:
[1] Patent: US6812238, 2004, B1. Location in patent: Page/Page column 21-22; 36
[2] Patent: CN106032356, 2016, A. Location in patent: Paragraph 0042; 0043
