624744-67-8
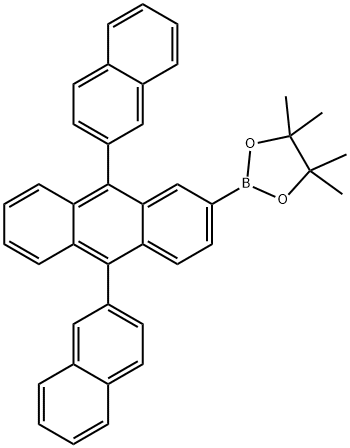 624744-67-8 结构式
624744-67-8 结构式
基本信息
9,10-双(2-萘基)蒽-2-硼酸频哪酯酯
9,10-双(2-萘基)-2-蒽硼酸频哪醇酯
2-硼酸频哪酯酯-9,10-双(2-萘基)蒽
9.10-双(2-萘基)蒽-2-硼酸频那醇脂
2-(9,10-二(萘-2-基)蒽-2-基)-4,4,5,5-四甲基-1,3,2-二氧杂硼烷
2-[9,10-二(萘-2-基)蒽-2-基]-4,4,5,5-四甲基-1,3,2-二氧杂环戊硼烷
9,10-di-2-naphthalenyl-2-anthracenylboronic acid bis(pinacol) ester
[9,10-Di(2-naphthyl)-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)]anthracene
2-(9,10-di(naphthalen-2-yl)anthracen-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
1,3,2-Dioxaborolane, 2-(9,10-di-2-naphthalenyl-2-anthracenyl)-4,4,5,5-tetramethyl-
2-[9,10-bis(naphthalen-2-yl)anthracen-2-yl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
物理化学性质
制备方法

474688-76-1
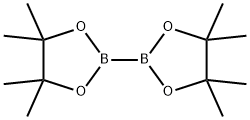
73183-34-3

624744-67-8
以2-溴-9,10-双(2-萘基)蒽(5.00 g,9.81 mmol)和联硼酸频那醇酯(2.75 g,10.8 mmol)为原料,在二恶烷(50 mL)中与乙酸钾(2.89 g,29.4 mmol)形成悬浮液。向该悬浮液中加入氯化钯(二苯基膦基二茂铁)(0.24 g,0.3 mmol)。将反应混合物在80℃下搅拌6小时,随后冷却至室温。反应完成后,用50 mL水稀释反应混合物,并用二氯甲烷(3×50 mL)进行萃取。合并有机相,用硫酸镁干燥,随后在减压下浓缩。粗产物用乙醇洗涤,并在真空下干燥,得到目标化合物2-(9,10-二(萘-2-基)蒽-2-基)-4,4,5,5-四甲基-1,3,2-二氧杂硼烷(5.46 g,收率92%)。质谱分析显示[M + H]+ = 557。
参考文献:
[1] Patent: WO2008/143440, 2008, A1. Location in patent: Page/Page column 70
[2] Patent: US7485733, 2009, B2. Location in patent: Page/Page column 79
[3] Patent: WO2003/95445, 2003, A1. Location in patent: Page/Page column 76
[4] Patent: WO2007/69847, 2007, A1. Location in patent: Page/Page column 28
[5] Patent: WO2008/13399, 2008, A1. Location in patent: Page/Page column 13
