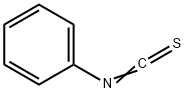
Phenyl isothiocyanate
- CAS No.
- 103-72-0
- Chemical Name:
- Phenyl isothiocyanate
- Synonyms
- PTC;Isothiocyanatobenzene;PITC;R1;Phenyl thiocyanate;Phenyl isothiocyanate,PITC;PHENYL ISOTHIOCYANATE SOLUTION, FOR PROT.SEQ. ANAL.;PIT;RR1;H174
- CBNumber:
- CB5733806
- Molecular Formula:
- C7H5NS
- Molecular Weight:
- 135.19
- MDL Number:
- MFCD00004798
- MOL File:
- 103-72-0.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| R1 NOTE: Both the cell line and DNA from the cell line may be available for this product. Please choose | CB_99043005 | 1 unit | $436 |
| R1 99043005, Reactivity: Anti-HLA-DQ | CB_99043005 | 1VIAL | $917 |
| Phenyl isothiocyanate for synthesis | 8.07028 | 100mL | $86.6 |
| Phenyl isothiocyanate for synthesis | 8.07028 | 250ML | $184 |
| Phenyl isothiocyanate reagent grade, 98% | 139742 | 100g | $81.3 |
| More product size | |||
| Melting point | −21 °C(lit.) |
|---|---|
| Boiling point | 218 °C(lit.) |
| Density | 1.132 g/mL at 20 °C(lit.) |
| vapor pressure | 10 hPa (20 °C) |
| refractive index |
n |
| Flash point | 190 °F |
| storage temp. | 2-8°C |
| solubility | water: insoluble |
| form | liquid |
| color | Clear pale yellow to yellow |
| Specific Gravity | 1.137 (20/4℃) |
| Water Solubility | Soluble in alcohol, and ether. Insoluble in water. |
| Sensitive | Moisture Sensitive |
| Merck | 14,7297 |
| BRN | 471392 |
| Henry's Law Constant | 3.3×10-3 mol/(m3Pa) at 25℃, Duchowicz et al. (2020) |
| Dielectric constant | 10.7(20℃) |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents, strong acids. |
| InChI | 1S/C7H5NS/c9-6-8-7-4-2-1-3-5-7/h1-5H |
| InChIKey | QKFJKGMPGYROCL-UHFFFAOYSA-N |
| SMILES | S=C=Nc1ccccc1 |
| LogP | 3.280 |
| Surface tension | 41.5mN/m at 20°C |
| CAS DataBase Reference | 103-72-0(CAS DataBase Reference) |
| FDA UNII | 0D58F84LSU |
| NIST Chemistry Reference | Benzene, isothiocyanato-(103-72-0) |
| EPA Substance Registry System | Benzene, isothiocyanato- (103-72-0) |
| UNSPSC Code | 12352209 |
| NACRES | NA.31 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS05,GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H314-H317-H334 | |||||||||
| Precautionary statements | P261-P270-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| PPE | Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter | |||||||||
| Hazard Codes | T,N,Xn,F | |||||||||
| Risk Statements | 25-34-42/43-51/53-67-65-50/53-36/37/38-22-11-38-63-23/24/25-36/38 | |||||||||
| Safety Statements | 9-16-29-33-60-61-62-36-26-23-45-36/37/39-38-28A-36/37 | |||||||||
| RIDADR | UN 1993 3/PG 2 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | NX9275000 | |||||||||
| F | 19 | |||||||||
| TSCA | TSCA listed | |||||||||
| HS Code | 2930 90 98 | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | II | |||||||||
| Storage Class | 6.1A - Combustible acute toxic Cat. 1 and 2 very toxic hazardous materials |
|||||||||
| Hazard Classifications | Acute Tox. 3 Oral Resp. Sens. 1 Skin Corr. 1B Skin Sens. 1 |
|||||||||
| Toxicity | LD50 orally in Rabbit: 157 mg/kg | |||||||||
| Limited Quantities | 100ml (liquid) or 0.5 Kg (solid) | |||||||||
| Excepted Quantities | Max Inner Pack (1g or 1ml) and Max Outer Pack (500g or 500ml) | |||||||||
| NFPA 704 |
|
Phenyl isothiocyanate price More Price(51)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | CB_99043005 | R1 NOTE: Both the cell line and DNA from the cell line may be available for this product. Please choose | 1 unit | $436 | 2026-04-30 | Buy | |
| Sigma-Aldrich | CB_99043005 | R1 99043005, Reactivity: Anti-HLA-DQ | 1VIAL | $917 | 2026-04-30 | Buy | |
| Sigma-Aldrich | 8.07028 | Phenyl isothiocyanate for synthesis | 103-72-0 | 100mL | $86.6 | 2026-04-30 | Buy |
| Sigma-Aldrich | 8.07028 | Phenyl isothiocyanate for synthesis | 103-72-0 | 250ML | $184 | 2026-04-30 | Buy |
| Sigma-Aldrich | 139742 | Phenyl isothiocyanate reagent grade, 98% | 103-72-0 | 100g | $81.3 | 2026-04-30 | Buy |
Phenyl isothiocyanate Chemical Properties,Uses,Production
Chemical Properties
colourless to pale yellow liquid with a penetrating odour
Uses
Phenyl isothiocyanate is the reagent of choice in automated Edman degradation systems. However, this reagent is highly toxic and for manual modification other reagents are preferred such as dimethy laminoazobenzene isothiocyanate (Chang, 1983; Wang et al, 2000) or trifluoroethyl isothiocyanate (Bartlet-Jones et al, 1994). This last reagent has the advantage of being volatile, so that the excess of reagent is easily removed by vacuum before MS analysis (Spengler, 1997). This compound was used successfully during seven successive cycles of manual cleavage coupled with MS analysis. Nevertheless, the high reactivity of such compounds seems to shows artefactual modification such as“acetylation' of the hydroxyl groups of the Ser and Thr. Allyl isothiocyanate shows better selectivity, but again this reagent is toxic and difficult to use in manual approaches (Gu & Preswich, 1997) .
Uses
Phenyl isothiocyanate acts as a derivatizing reagent for primary and secondary amines. It is used in sequencing peptides by Edman degradation and in amino acid analyses by HPLC. It is used for derivatizing N-terminal amino acids of proteins for automated sequential analysis. It is a synthon for dithiadiazafulvalenes.
Definition
ChEBI: Phenyl isothiocyanate is an isothiocyanate having a phenyl group attached to the nitrogen; used for amino acid sequencing in the Edman degradation. It has a role as an allergen and a reagent.
Preparation
Synthetic procedure for phenyl isothiocyanate in 1-mol scale
Into a 2-L jacketed flask, 91.2 g of CS2 (1.2 mol) was dropwise added to a mixture of aniline (93.0 g, 1.0 mol) and K2CO3 (276.0 g, 2.0 mol) in 700 mL of water at room temperature within a period of 2.5 h. After the addition was complete, the mixture was stirred another 2 h. Then, the mixture was cooled to 0 °C, and a solution of 92.3 g of TCT (0.5 mol) in 450 mL of CH2Cl2 was dropwise added within 4 h. After the addition was complete, the mixture was stirred another 1 h to complete the conversion. The resulting mixture was basified to pH >11 with 250 mL of 6 N NaOH and yielded a clear solution. The organic layer was separated and the aqueous layer was extracted with 150 mL of CH2Cl2. The combined organic layers were dried over anhydrous Na2SO4, filtered, and the solvent of filtrate was removed via distillation under atmospheric pressure with a 25-cm Vigreux column. The residue was vacuum distilled and the desired product fraction was collected at 72–74 °C/1 mmHg. Finally, 127.0 g of colorless liquid (94%) was obtained.
Biological Functions
Phenyl isothiocyanate (PITC) is a well-established reagent in protein chemistry since its introduction in Edman degradation. PITC reacts with primary and secondary amines under alkaline conditions within 20 min. The resulting phenylthiocarbamyl (PTC) derivatives of the amino acids are stable and do not interfere with reaction by-products during chromatography. The absorption maximum is around 245 nm with a detection limit of 1 pmol.
Phenyl isothiocyanate may be employed as a derivatization reagent for high-performance liquid chromatographic (HPLC) analysis of various amphetamine derivatives in body fluids for forensic purposes.
Synthesis Reference(s)
Tetrahedron Letters, 38, p. 1597, 1997 DOI: 10.1016/S0040-4039(97)00121-4
Synthesis, p. 300, 1989 DOI: 10.1055/s-1989-27231
Tetrahedron Letters, 23, p. 447, 1982 DOI: 10.1016/S0040-4039(00)86856-2
General Description
Phenyl isothiocyanate is an aromatic isothiocyanate. It participates in dehydration reactions of alcohols. It is widely used for synthesis of various biologically important heterocyclic compounds.
Purification Methods
It is insoluble in H2O, but soluble in Et2O and EtOH. If impure (due to formation of thiourea), then steam distil it into a receiver containing 5-10mL of N H2SO4. Separate the oil, dry over CaCl2 and distil it under vacuum. [Dains et al. Org Synth Coll Vol I 447 1941, Beilstein 12 IV 867.]
Phenyl isothiocyanate Preparation Products And Raw materials
Raw materials
Preparation Products
1of2
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Changzhou Zechong Biopharma Co., Ltd. | +undefined18621330623 | sales@zechongchem.com | China | 28 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-86-13131129325 +8613131129325 | sales1@chuanghaibio.com | China | 5235 | 58 |
| Capot Chemical Co.,Ltd. | +86-(0)57185586718; +8613336195806 | sales@capot.com | China | 29640 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21590 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29819 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| Neostar United (Changzhou) Industrial Co., Ltd. | +86-0519-85551759 +8613506123987 | marketing1@neostarunited.com | China | 8828 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34563 | 58 |
| Shanghai UCHEM Inc. | +862156762820 +86-13564624040 | sales@myuchem.com | China | 8736 | 58 |
| AFINE CHEMICALS LIMITED | +86-0571-85134551 | sales@afinechem.com | China | 15082 | 58 |
View Lastest Price from Phenyl isothiocyanate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2025-05-26 | Phenyl isothiocyanate
103-72-0
|
US $5.00-2.00 / KG | 1KG | 99% | 10000kg | Hebei Chuanghai Biotechnology Co,.LTD | |
 |
2021-07-02 | PHENYL ISOTHIOCYANATE
103-72-0
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd | |
 |
2019-07-06 | PHENYL ISOTHIOCYANATE
103-72-0
|
US $11.00 / KG | 1KG | 99% | 1000KG | Career Henan Chemical Co |
-

- Phenyl isothiocyanate
103-72-0
- US $5.00-2.00 / KG
- 99%
- Hebei Chuanghai Biotechnology Co,.LTD
-

- PHENYL ISOTHIOCYANATE
103-72-0
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd
-

- PHENYL ISOTHIOCYANATE
103-72-0
- US $11.00 / KG
- 99%
- Career Henan Chemical Co
103-72-0(Phenyl isothiocyanate)Related Search:
1of4