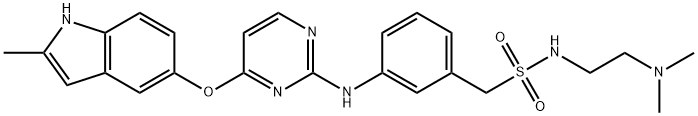
Surufatinib
- CAS No.
- 1308672-74-3
- Chemical Name:
- Surufatinib
- Synonyms
- HMPL012;CS-2346;HMPL-012;KDR-IN-1;EOS-61498;Surufatinib;Surufatinib(HPML-012);Sulfatinib, 10 mM in DMSO;HMPL-012;SURUFATINIB;HMPL012;N-(2-(DIMETHYLAMINO)ETHYL)-1-(3-((4-((2-METHYL-1H-INDOL-5-YL)OXY)PYRIMIDIN-2-YL)AMINO)PHENYL)METHANE
- CBNumber:
- CB72711244
- Molecular Formula:
- C24H28N6O3S
- Molecular Weight:
- 480.58
- MDL Number:
- MOL File:
- 1308672-74-3.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| Sulfatinib | S688948 | 1mg | $60 |
| Sulfatinib | S688948 | 10mg | $340 |
| Sulfatinib 98.01% | CS-5949 | 100mg | $450 |
| Sulfatinib 98.01% | CS-5949 | 5mg | $90 |
| Sulfatinib 98.01% | CS-5949 | 10mg | $120 |
| More product size | |||
| Boiling point | 712.9±70.0 °C(Predicted) |
|---|---|
| Density | 1.332±0.06 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMSO:98.0(Max Conc. mg/mL);203.92(Max Conc. mM) |
| form | A solid |
| pka | 5.22±0.40(Predicted) |
| color | White to off-white |
| InChIKey | TTZSNFLLYPYKIL-UHFFFAOYSA-N |
| SMILES | C1(CS(NCCN(C)C)(=O)=O)=CC=CC(NC2=NC=CC(OC3C=CC4=C(C=3)C=C(C)N4)=N2)=C1 |
| FDA UNII | B2K5L1L8S9 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302-H315-H319-H335 |
| Precautionary statements | P261-P305+P351+P338 |
Surufatinib price
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| TRC | S688948 | Sulfatinib | 1308672-74-3 | 1mg | $60 | 2021-12-16 | Buy |
| TRC | S688948 | Sulfatinib | 1308672-74-3 | 10mg | $340 | 2021-12-16 | Buy |
| ChemScene | CS-5949 | Sulfatinib 98.01% | 1308672-74-3 | 100mg | $450 | 2021-12-16 | Buy |
| ChemScene | CS-5949 | Sulfatinib 98.01% | 1308672-74-3 | 5mg | $90 | 2021-12-16 | Buy |
| ChemScene | CS-5949 | Sulfatinib 98.01% | 1308672-74-3 | 10mg | $120 | 2021-12-16 | Buy |
Surufatinib Chemical Properties,Uses,Production
Uses
Sulfatinib is used in pharmaceutical compositions comprising of micronized drug.
Mechanism of action
Surufatinib can inhibit multiple targets related to angiogenesis and tumor growth, including VEGFR, FGFR1 and CSF-1R.
Synthesis
Treatment of sulfonyl chloride 358 at room temperature using triethylamine and 2,2,2-trifluoroethanol (TFE) as solvents followed by hydrogenation afforded aniline 360. Exposure of 360 to DBU under microwave conditions generated an intermediate sulfinyl group. This intermediate sulfinyl group subsequently underwent nucleophilic attack by a commercial diamine, of which diamine 361 afforded the desired sulfonamide 362. Dichloropyrimidine 363 underwent a substitution reaction with hydroxyindole 364 to afford diaryl ether 365. Diaryl ether 365 then underwent a second substitution reaction with amine 362 under acidic conditions. Exposure of the product to a base in aqueous solution ultimately afforded surufatinib in 50% yield.
in vivo
In animal studies, a single oral dosing of Sulfatinib inhibits VEGF stimulated VEGFR2 phosphorylation in lung tissues of nude mice in an exposure-dependent manner. Furthermore, elevation of FGF23 levels in plasma 24 hours post dosing suggests suppression of FGFR signaling. Sulfatinib demonstrates potent tumor growth inhibition in multiple human xenograft models and decreases CD31 expression remarkably, suggesting strong inhibition on angiogenesis through VEGFR and FGFR signaling. In a syngeneic murine colon cancer model CT-26, Sulfatinib demonstrates moderate tumor growth inhibition after single agent treatment[1]. After oral dosing of 10 mg/kg, the AUC and Cmax are 397 ng/mL and 138ng/mL in the mouse, respectively[1].
IC 50
VEGFR1; VEGFR2; VEGFR3; FGFR1; CSF1R


Surufatinib Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 33024 | 60 |
| Jinan Carbotang Biotech Co.,Ltd. | +8615866703830 | figo.gao@foxmail.com | China | 8497 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354; +17819995354 | marketing@targetmol.com | United States | 32467 | 58 |
| Hubei Ipure Biology Co., Ltd | +8613367258412 | ada@ipurechemical.com | China | 10236 | 58 |
| HONG KONG IPURE BIOLOGY CO.,LIMITED | 86 18062405514 18062405514 | ada@ipurechemical.com | CHINA | 3461 | 58 |
| HANGZHOU CLAP TECHNOLOGY CO.,LTD | 86-571-88216897,88216896 13588875226 | sales@hzclap.com | CHINA | 6312 | 58 |
| InvivoChem | +1-708-310-1919 +1-13798911105 | sales@invivochem.cn | United States | 6391 | 58 |
| HANGZHOU LEAP CHEM CO., LTD. | +86-571-87711850 | market18@leapchem.com | China | 24597 | 58 |
| Nantong HI-FUTURE Biology Co., Ltd. | +undefined18051384581 | sales@chemhifuture.com | China | 3135 | 58 |
| Wuhan Topule Biopharmaceutical Co., Ltd | +8618327326525 | masar@topule.com | China | 8467 | 58 |
Related articles
- Synthesis of Surufatinib
- Surufatinib is synthesised from dichloropyrimidine and hydroxyindole by substitution reaction. This process requires the parti....
- Jan 5,2024
View Lastest Price from Surufatinib manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-17 | Sulfatinib
1308672-74-3
|
US $39.00-97.00 / mg | 99.97% | 10g | TargetMol Chemicals Inc. |
-

- Sulfatinib
1308672-74-3
- US $39.00-97.00 / mg
- 99.97%
- TargetMol Chemicals Inc.





