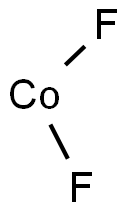
Cobalt(II) fluoride
- CAS No.
- 10026-17-2
- Chemical Name:
- Cobalt(II) fluoride
- Synonyms
- difluorocobalt;COBALT FLUORIDE;Cobalt(Ⅱ)fluorde;Cobalt(Ⅱ)fluoride;COBALT DIFLUORIDE;COBALTOUS FLUORIDE;COBALT(II) FLUORIDE;Two cobalt fluoride;COBALT FLUORIDE PINK;cobaltfluoride(cof2)
- CBNumber:
- CB7681660
- Molecular Formula:
- CoF2
Lewis structure
 Crystal structure
Crystal structure
- Molecular Weight:
- 96.93
- MDL Number:
- MFCD00010941
- MOL File:
- 10026-17-2.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Cobalt(II) fluoride powder | 236128 | 25g | $112 |
| Cobalt(II) fluoride, anhydrous, 99% | 93-2738 | 10g | $46 |
| Cobalt(II) fluoride, anhydrous, 99% | 93-2738 | 50g | $164 |
| COBALT(II) FLUORIDE ANHYDROUS 95.00% | ING0003978 | 25G | $1079.93 |
| COBALT(II) FLUORIDE ANHYDROUS 95.00% | ING0003978 | 100G | $2431.28 |
| More product size | |||
| Melting point | 1200 °C | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boiling point | 1400 °C | ||||||||||||||
| Density | 4.46 g/mL at 25 °C | ||||||||||||||
| solubility | soluble in acid solutions | ||||||||||||||
| form | powder | ||||||||||||||
| color | Pink | ||||||||||||||
| Specific Gravity | 4.46 | ||||||||||||||
| Water Solubility | Soluble in water, alcohol and acetone. | ||||||||||||||
| Crystal Structure | Rutile type | ||||||||||||||
| Sensitive | Hygroscopic | ||||||||||||||
| Merck | 14,2440 | ||||||||||||||
| crystal system | square | ||||||||||||||
| Space group | P42/mnm | ||||||||||||||
| Lattice constant |
|
||||||||||||||
| Exposure limits |
ACGIH: TWA 0.02 mg/m3; TWA 2.5 mg/m3 NIOSH: IDLH 250 mg/m3; TWA 2.5 mg/m3 |
||||||||||||||
| Stability | hygroscopic | ||||||||||||||
| Major Application | battery manufacturing | ||||||||||||||
| InChI | 1S/Co.2FH/h;2*1H/q+2;;/p-2 | ||||||||||||||
| InChIKey | YCYBZKSMUPTWEE-UHFFFAOYSA-L | ||||||||||||||
| SMILES | F[Co]F | ||||||||||||||
| CAS DataBase Reference | 10026-17-2(CAS DataBase Reference) | ||||||||||||||
| EWG's Food Scores | 1 | ||||||||||||||
| FDA UNII | 9KI67810UR | ||||||||||||||
| EPA Substance Registry System | Cobalt fluoride (CoF2) (10026-17-2) | ||||||||||||||
| UNSPSC Code | 12352302 | ||||||||||||||
| NACRES | NA.23 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H314 | |||||||||
| Precautionary statements | P260-P270-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| PPE | Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges, type P3 (EN 143) respirator cartridges | |||||||||
| Hazard Codes | T,C | |||||||||
| Risk Statements | 25-34 | |||||||||
| Safety Statements | 26-36/37/39-45-50A-28A | |||||||||
| RIDADR | UN 2923 8/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | GG0770000 | |||||||||
| F | 21 | |||||||||
| Hazard Note | Toxic | |||||||||
| TSCA | TSCA listed | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 28261990 | |||||||||
| Storage Class | 6.1A - Combustible acute toxic Cat. 1 and 2 very toxic hazardous materials |
|||||||||
| Hazard Classifications | Acute Tox. 3 Oral Skin Corr. 1B |
|||||||||
| Hazardous Substances Data | 10026-17-2(Hazardous Substances Data) | |||||||||
| Limited Quantities | 5.0 L (1.3 gallons) (liquid) or 5.0 kg (11 lbs) (solid) | |||||||||
| Excepted Quantities | Max Inner Pack (30g or 30ml) and Max Outer Pack (1Kg or 1L) | |||||||||
| NFPA 704 |
|
Cobalt(II) fluoride price More Price(24)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 236128 | Cobalt(II) fluoride powder | 10026-17-2 | 25g | $112 | 2023-06-20 | Buy |
| Strem Chemicals | 93-2738 | Cobalt(II) fluoride, anhydrous, 99% | 10026-17-2 | 10g | $46 | 2024-03-01 | Buy |
| Strem Chemicals | 93-2738 | Cobalt(II) fluoride, anhydrous, 99% | 10026-17-2 | 50g | $164 | 2024-03-01 | Buy |
| American Custom Chemicals Corporation | ING0003978 | COBALT(II) FLUORIDE ANHYDROUS 95.00% | 10026-17-2 | 25G | $1079.93 | 2021-12-16 | Buy |
| American Custom Chemicals Corporation | ING0003978 | COBALT(II) FLUORIDE ANHYDROUS 95.00% | 10026-17-2 | 100G | $2431.28 | 2021-12-16 | Buy |
Cobalt(II) fluoride Chemical Properties,Uses,Production
Preparation
Cobalt(II) fluoride is prepared by heating anhydrous cobalt(II) chloride or oxide in a stream of hydrogen fluoride:
CoCl2 + 2HF → CoF2 + 2HCl
CoO + 2HF → CoF2 + 2H2O
Also, cobalt(II) fluoride can be prepared as a tetrahydrate, CoF2•4H2O by dissolving cobalt(II) hydroxide in hydrofluoric acid. The tetrahydrate is then dehydrated to anhydrous fluoride. Elemental fluorine combines with cobalt at 450°C forming mixtures of cobalt(II)–and cobalt(III) fluorides.
Chemical Properties
Rose-red crystals or powder. Soluble in cold water and hydrofluoric acid. Decomposes in hot water. Ammine complexes can be prepared from the hydrate.
Physical properties
Red tetragonal crystal; density 4.46 g/cm3; melts at 1,127°C; vaporizes around 1,400°C; sparingly soluble in water; soluble in warm mineral acids; decomposes in boiling water. Tetrahydrate is red orthogonal crystal; density 2.22 g/cm3; decomposes on heating; soluble in water; di- and trihydrates are soluble in water.
Uses
Cobalt(II) fluoride can be used as a catalyst to alloy metals. It is also used for optical deposition, of which it tremendously improves optical quality. The compound may be used in dental care.
Uses
Catalyst for organic reactions.
General Description
Violet to red solid. Sinks and mixes slowly with water.
Reactivity Profile
Cobalt(II) fluoride is a toxic, light brown crystalline material, when exposed to water Cobalt(II) fluoride forms highly corrosive hydrofluoric acid. When heated to decomposition Cobalt(II) fluoride emits toxic fumes of metallic cobalt and fluorides [Lewis, 3rd ed., 1993, p. 344].
Hazard
Highly toxic.
Health Hazard
INHALATION: Inhalation of dust may cause pulmonary symptoms. EYES: Irritation. SKIN: Skin rashes, dermatitis. INGESTION: Nausea and vomiting caused by local irritation.
LD50 oral (rat): 150 mg/kg
Safety Profile
Poison by ingestion. When heated to decomposition it emits toxic fumes of Co and F-.
Cobalt(II) fluoride Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21592 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29795 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34563 | 58 |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 | eric@witopchemical.com | China | 23541 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +86-86-17392712779 +8617392712697 | 1022@dideu.com | China | 8401 | 58 |
| GLR Innovations | +91 9891111994 | info@glrgroup.in | India | 4535 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615531151365 | mina@chuanghaibio.com | China | 18126 | 58 |
| GIHI CHEMICALS CO.,LIMITED | +86-571-86217390; +8618058761490 | info@gihichemicals.com | China | 49968 | 58 |
| SUZHOU SENFEIDA CHEMICAL CO.,LTD | +86-0512-83500002 +8618662433356 | 3899766280@qq.com | China | 26362 | 58 |
| Aladdin Scientific | tp@aladdinsci.com | United States | 57505 | 58 |
View Lastest Price from Cobalt(II) fluoride manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2020-01-10 | Cobalt(II) fluoride
10026-17-2
|
US $1.00 / KG | 1g | 98% | 200kgs | Career Henan Chemical Co |
-

- Cobalt(II) fluoride
10026-17-2
- US $1.00 / KG
- 98%
- Career Henan Chemical Co