- Methyl Methylcarbamate
-

- $100.00 / 1KG
-
2026-02-02
- CAS:6642-30-4
- Min. Order: 1KG
- Purity: 98%HPLC
- Supply Ability: 100tons
- methyl methylcarbamate
-

- $1.00 / 1KG
-
2020-01-13
- CAS:6642-30-4
- Min. Order: 1KG
- Purity: 85.0-99.8%
- Supply Ability: 200kgs
|
| | methyl methylcarbamate Basic information |
| Product Name: | methyl methylcarbamate | | Synonyms: | methyl methylcarbamate;Methylcarbamic acid methyl ester;N-Methylcarbamic acid methyl ester;Nsc15396;CarbaMic acid,N-Methyl-, Methyl ester;methyl N-methylcarbamate | | CAS: | 6642-30-4 | | MF: | C3H7NO2 | | MW: | 89.09 | | EINECS: | 229-661-5 | | Product Categories: | | | Mol File: | 6642-30-4.mol | 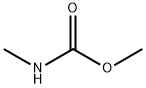 |
| | methyl methylcarbamate Chemical Properties |
| Melting point | 173-176 °C | | Boiling point | 68 °C(Press: 17 Torr) | | density | 0.986±0.06 g/cm3(Predicted) | | pka | 12.77±0.46(Predicted) |
| | methyl methylcarbamate Usage And Synthesis |
| Uses | Methyl Methylcarbamate is used to study the catalytic metathesis of N-Methylformamide. | | Synthesis | Step 1: Preparation of dibutyltin oxide (DBTO) Bu2SnCl2 (9.48 g, 31.2 mmol) was dissolved in 30
mL of ether, stirred, and left to stand. To a 100 mL fitted with a thermometer, stirring distillation apparatus, and a dropping funnel
In a three-necked flask, add 30 mL of 10% NaOH aqueous solution by mass, heat to 323 K, and slowly drop in the above clarified ether solution to be used with stirring, while using the distillation device to recover the evaporated ether, and after dropping, change the reflux device to boil the solution for 0.5 h. Filter out the white ether solution and remove it from the flask, then remove the ether from the flask.
0.5 h. The white precipitate was filtered out, then washed with distilled water to neutral, and dried to obtain white granular solid (yield >98%). Step II: Preparation of methyl N-methylcarbamate (MMC) 1,3-dimethylurea ( 3.04 g, 34.50 mmol), dibutyltin oxide ( 0. 56 g, 2. 25 mmol) and 28.76g
DMC were added to a 50 mL stainless steel reactor.DMC was used as both reactant and solvent. The reaction was stirred at 423 K for 4 h. The reaction was cooled down to room temperature.
temperature and separated on a column to give a light yellow liquid MMC in 99% yield and 96% chromatographic purity. |
| | methyl methylcarbamate Preparation Products And Raw materials |
|