|
|
| | (S)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]-ETHYLDI-TERT.-BUTYLPHOSPHINE Basic information | | Reaction |
| Product Name: | (S)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]-ETHYLDI-TERT.-BUTYLPHOSPHINE | | Synonyms: | 1,2,3,4,5-Cyclopentanepentayl, compd. with 1-[(1S)-1-[bis(1,1-dim ethylethyl)phosphino]ethyl]-2-(diphenylphosphino)-1,2,3,4,5-cyclo pentanepentayl, iron salt (1:1:1);(2S)-1-[(1S)-1-[Bis(1,1-dimethylethyl)phosphino]ethyl]-2-(diphenylphosphino)ferrocene;(S)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]-ETHYLDI-TERT.-BUTYLPHOSPHINE;(S)-(+)-1-[(R)-2-(Diphenylphosphino)ferrocenyl]ethyldi-t-butylphosphine,min.97%;(s,s)-1-[1-(di-tert-butylphosphino)ethyl]-2-(diphenylphosphino]ferrocene (acc to cas);(S)-(+)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]ETHYLDI-T-BUTYLPHOSPHINE, MIN. 97%;(S)-1-[(RP)-2-(Diphenylphosphino)ferrocenyl]ethyldi-tert-butylphosphine;Josiphos SL-J002-2, (2S)-1-[(1S)-1-[Bis(1,1-dimethylethyl)phosphino]ethyl]-2-(diphenylphosphino)ferrocene (acc to CAS) | | CAS: | 277306-29-3 | | MF: | C32H40FeP2 | | MW: | 542.45 | | EINECS: | | | Product Categories: | Ferrocene Series;Josiphos Series;Chiral Phosphine;organophosphine ligand | | Mol File: | 277306-29-3.mol | ![(S)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]-ETHYLDI-TERT.-BUTYLPHOSPHINE Structure](CAS/GIF/277306-29-3.gif) |
| | (S)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]-ETHYLDI-TERT.-BUTYLPHOSPHINE Chemical Properties |
| alpha | +412° ±15° (c 0.5, CHCl3) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | form | Powder | | color | orange | | Optical Rotation | [α]20/D +410±15°, c = 0.5 in chloroform | | InChI | 1S/C27H35P2.C5H5.Fe/c1-21(29(26(2,3)4)27(5,6)7)24-19-14-20-25(24)28(22-15-10-8-11-16-22)23-17-12-9-13-18-23;1-2-4-5-3-1;/h8-21H,1-7H3;1-5H;/t21-;;/m0../s1 | | InChIKey | STAGMXYQPGLLTD-FGJQBABTSA-N | | SMILES | [Fe].[CH]1[CH][CH][CH][CH]1.C[C@@H]([C]2[CH][CH][CH][C]2P(c3ccccc3)c4ccccc4)P(C(C)(C)C)C(C)(C)C |
| Safety Statements | 22-24/25 | | WGK Germany | 3 | | F | 10 | | HS Code | 29319090 | | Storage Class | 11 - Combustible Solids |
| | (S)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]-ETHYLDI-TERT.-BUTYLPHOSPHINE Usage And Synthesis |
| Reaction |
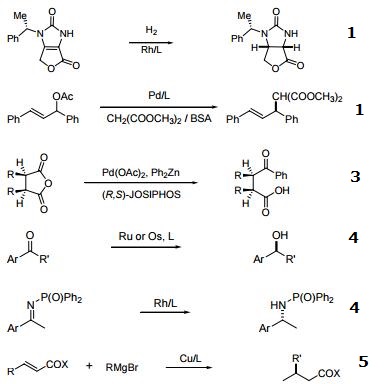
- Ferrocenylphosphine ligands of the type cpFecp(PR2)(*CH(CH3)PR'2) are a class of asymmetric ligands developed at Solvias in Basel, Switzerland . Ligands of this type are currently used industrially in the stereoselective synthesis of commercial products. A unique feature of these bidentate ligands is the presence of a fixed phosphine moiety and a stereogenic, functionalized side chain, which can be easily modified to accommodate electronic and steric requirements. Based on a versatile synthetic procedure starting with optically active ferrocenes of the type cpFecp(PR2)(*CH(CH3)X) [X = OAc or NR2], a variety of donor atoms can be introduced into the side chain. These ferrocene based phosphine ligands have wide application in the stereoselective hydrogenation of substituted acetamidoacrylates, enol acetates, β-ketoesters and simple alkenes.
- Useful as a ligand in Pd-catalyzed C-N bond-forming reactions.
- Pd-catalyzed enantioselective alkylative desymmetrization of meso-succinic anhydrides.
- Asymmetric hydrogenation of ketones and phosphinylketimines.
- Michael addition of Grignard reagents to ",$-unsaturated esters and thioesters.
- Boration of ",$-unsaturated esters and nitriles.
- Reaction of aryl halides with ammonia.
- Cu-catalyzed reduction of activated C=C bonds with PMHS.
- Regio- and enantioselective hydroboration of vinyl arenes.
- Rh-catalyzed asymmetric ring-opening reactions of oxabicyclic alkenes.
- 1,2-Migrations in Pd-catalyzed Negishi couplings with JosiPhos ligands.
| | Chemical Properties | Orange powder | | Uses | (S)-1-[(RP)-2-(Diphenylphosphino)ferrocenyl]ethyldi-tert-butylphosphine is β-boration catalyst; used in preparation of Sitagliptin β-Amino-2,4,5-trifluorobenzenebutanoic Acid derivatives intermediates via Grignard exchange reaction, cross coupling, β-boration, oxidation and Amination. | | General Description | sold in collaboration with Solvias AG |
| | (S)-1-[(R)-2-(DIPHENYLPHOSPHINO)FERROCENYL]-ETHYLDI-TERT.-BUTYLPHOSPHINE Preparation Products And Raw materials |
|