Apafant manufacturers
- Apafant
-

- $56.00 / 1mg
-
2026-05-11
- CAS:105219-56-5
- Min. Order:
- Purity: 99.56%
- Supply Ability: 10g
- Apafant
-

- $56.00 / 1mg
-
2026-05-11
- CAS:105219-56-5
- Min. Order:
- Purity: 99.56%
- Supply Ability: 10g
- Apafant
-

- $1.00 / 1KG
-
2024-10-22
- CAS:105219-56-5
- Min. Order: 1KG
- Purity: MIn98%HPLC/LC
- Supply Ability: 100KGS
|
| | Apafant Basic information |
| Product Name: | Apafant | | Synonyms: | 4-[3-[4-(2-Chlorophenyl)-9-methyl-6H-thieno[3,2-f][1,2,4]triazolo[4,3-a][1,4]diazepin-2-yl]-1-oxopropyl]-morpholine;WEB 208;WEB 2086BS;4-[3-[4-(2-Chlorophenyl)-9-methyl-6H-thieno[3,2-f][1,2,4]triazolo[4,3-α][1,4]diazepin-2-yl]-1-oxopropyl]-morpholine;3-[4-(2-Chlorophenyl)-9-methyl-6H-thieno[3,2-f][1,2,4]triazolo[4,3-a][1,4]diazepin-2-yl]-1-(4-morpholinyl)-1-propanone;4-[3-[4[(2-Chlorophenyl)-9-methyl-6H-thieno[3,2-f][1,2,4]triazolo[4,3-a]diazepin-2-yl]-1-oxopropyl]morpholine;4-(2-Chlorophenyl)-9-methyl-2-(3-morpholino-3-oxopropyl)-6H-thieno[3,2- f] [1,2,4]triazolo[4,3-a][1,4]diazepine;4-[3-[4[(2-CHLOROPHENYL)-9-METHYL-6H-THIENO[3,2-F][1,2,4]TRIAZOLO[4,3-A]DIAZEPIN-2-YL]-1-OXOPROPYL]]MORPHOLINE | | CAS: | 105219-56-5 | | MF: | C22H22ClN5O2S | | MW: | 455.96 | | EINECS: | | | Product Categories: | PAF receptor;Intermediates & Fine Chemicals;Pharmaceuticals;Sulfur & Selenium Compounds | | Mol File: | 105219-56-5.mol | 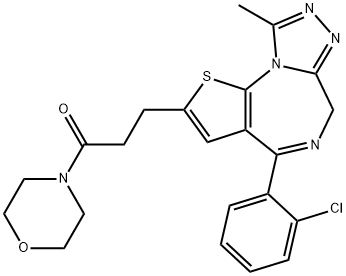 |
| | Apafant Chemical Properties |
| Melting point | 186-188°C | | Boiling point | 720.2±70.0 °C(Predicted) | | density | 1.48 | | storage temp. | 2-8°C | | solubility | DMSO: soluble10mg/mL (clear solution) | | form | powder | | pka | 2.36±0.40(Predicted) | | color | white to beige | | Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 3 months. | | InChI | 1S/C22H22ClN5O2S/c1-14-25-26-19-13-24-21(16-4-2-3-5-18(16)23)17-12-15(31-22(17)28(14)19)6-7-20(29)27-8-10-30-11-9-27/h2-5,12H,6-11,13H2,1H3 | | InChIKey | JGPJQFOROWSRRS-UHFFFAOYSA-N | | SMILES | Cc1nnc2CN=C(c3ccccc3Cl)c4cc(CCC(=O)N5CCOCC5)sc4-n12 |
| WGK Germany | 3 | | Storage Class | 11 - Combustible Solids | | Toxicity | LD50 in mice (mg/kg): 540 i.v.; 4600 orally (Casals-Stenzel) |
| | Apafant Usage And Synthesis |
| Description | WEB-2086 (105219-56-5) is a potent and selective antagonist of the platelet activating factor (PAF) receptor (Ki = 16.3 nM).1Displays an activity profile which includes antiinflammatory, antiangiogenesis and anticancer activity.2Along with leukotriene antagonists, WEB-2086 cooperatively provides a robust antiinflammatory effect regulating PMNL migration and edema formation.3Displays analgesic effects in animal models of neuropathic pain.4Active in vivo. | | Chemical Properties | White Solid | | Uses | Platelet activating factor (PAF) antagonist. | | Uses | Tool to evaluate the role of PAF in experimental models of human disease. | | Definition | ChEBI: LSM-2613 is an organonitrogen heterocyclic compound and an organosulfur heterocyclic compound. | | Biological Activity | Potent, selective platelet-activating factor (PAF) receptor antagonist (K i = 16.3 nM). Displays anti-inflammatory, antiangiogenic and anticancer activity. Inhibits growth and proliferation of MCF-7 breast cancer cells. | | Biochem/physiol Actions | WEB2086 is a very potent, selective platelet-activating factor (PAF) antagonist (IC50 = 16 nM). WEB2086 inhibits the proliferation of tumor cells and slows tumor growth in xenograft tumor models, and has been shown to inhibit angiogenesis. | | storage | Desiccate at +4°C | | References | [1] GORDON DENT . Characterization of PAF receptors on human neutrophils using the specific antagonist, WEB 2086 Correlation between receptor binding and function[J]. FEBS Letters, 1989, 244 2: Pages 365-368. DOI:10.1016/0014-5793(89)80564-2
[2] C CELLAI. Growth inhibition and differentiation of human breast cancer cells by the PAFR antagonist WEB-2086[J]. British Journal of Cancer, 2006, 94 11: 1637-1642. DOI:10.1038/sj.bjc.6603156
[3] CLAUDIA S. BITENCOURT. Cooperative role of endogenous leucotrienes and platelet-activating factor in ischaemia–reperfusion-mediated tissue injury[J]. JOURNAL OF CELLULAR AND MOLECULAR MEDICINE, 2013, 17 12: 1554-1565. DOI:10.1111/jcmm.12118
[4] N. MOTOYAMA. Pain-releasing action of Platelet-activating factor (PAF) antagonists in neuropathic pain animal models and the mechanisms of action[J]. European Journal of Pain, 2013, 17 8: 1156-1167. DOI:10.1002/j.1532-2149.2013.00289.x |
| | Apafant Preparation Products And Raw materials |
|