|
|
| | N-Boc-L-homoserine Methyl Ester Basic information |
| Product Name: | N-Boc-L-homoserine Methyl Ester | | Synonyms: | methyl (2S)-4-hydroxy-2-[(2-methylpropan-2-yl)oxycarbonylamino]butanoate;N-[(1,1-Dimethylethoxy)carbonyl]-L-homoserine Methyl Ester;N-Boc-L-homoserine Methyl Ester;(S)-2-(tert-Butoxycarbonylamino)-4-hydroxybutyric Acid Methyl Este;methyl (2S)-2-{[(tert-butoxy)carbonyl]amino}-4-hydroxybutanoate;L-Homoserine, N-[(1,1-dimethylethoxy)carbonyl]-, methyl ester;tert-butyl (S)-1-(methoxycarbonyl)-3-hydroxypropylcarbamate;Boc-L-Homoserine methyl ester | | CAS: | 120042-11-7 | | MF: | C10H19NO5 | | MW: | 233.26 | | EINECS: | | | Product Categories: | | | Mol File: | 120042-11-7.mol | 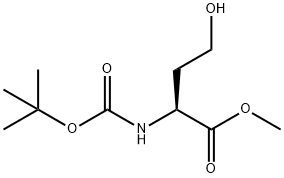 |
| | N-Boc-L-homoserine Methyl Ester Chemical Properties |
| Boiling point | 364.6±37.0 °C(Predicted) | | density | 1.125±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,Store in freezer, under -20°C | | pka | 10.91±0.46(Predicted) | | Appearance | Colorless to light yellow Viscous Liquid |
| | N-Boc-L-homoserine Methyl Ester Usage And Synthesis |
| Synthesis | To a solution of (S)-2-(tert-butoxycarbonylamino)-4-hydroxybutyric acid 206 (8.5 g, 38 mmol) in DMF (50 mL) was added K2CO3 (5.24 g, 38 mmol) and iodomethane (5.4 g, 38 mmol). The reaction mixture was stirred at room temperature overnight. After the reaction was completed, the reaction mixture was diluted with water (500 mL) and extracted with dichloromethane (500 mL). The organic layers were combined, dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure to remove the solvent to afford the crude product (S)-2-((tert-butoxycarbonyl)amino)-4-hydroxybutyric acid methyl ester 207 (8.5 g, 90% yield). | | References | [1] Patent: WO2017/189866, 2017, A1. Location in patent: Paragraph 0243
[2] Patent: WO2017/156074, 2017, A1. Location in patent: Paragraph 00494
[3] Biochemistry, 2017, vol. 56, # 8, p. 1062 - 1074
[4] Patent: WO2016/112088, 2016, A1. Location in patent: Paragraph 0481 |
| | N-Boc-L-homoserine Methyl Ester Preparation Products And Raw materials |
|