- Teprenone
-

- $98.00 / 500mg
-
2026-04-09
- CAS:6809-52-5
- Min. Order:
- Purity: 99.10%
- Supply Ability: 10g
- Teprenone
-

- $0.00 / 1kg
-
2026-03-05
- CAS:6809-52-5
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 20tons
- Teprenone
-
- $35.00 / 1kg
-
2025-09-25
- CAS:6809-52-5
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | Teprenone Basic information |
| Product Name: | Teprenone | | Synonyms: | All trans-Teprenone;(5Z,9Z,13Z)-6,10,14,18-tetramethyl-2-nonadeca-5,9,13,17-tetraenone;6,10,14,18-tetramethyl-5,9,13,17-nonadecatetraen-2-one;TEPRENONE;13,17-nonadecatetraen-2-one,6,10,14,18-tetramethyl-9;geranylgeranylacetone;6,10,14,18-Tetramethyl-5,9,13,17-nonadecatetren-2-one;Selbex | | CAS: | 6809-52-5 | | MF: | C23H38O | | MW: | 330.55 | | EINECS: | 614-276-0 | | Product Categories: | Aliphatics;Intermediates & Fine Chemicals;Pharmaceuticals;6809-52-5 | | Mol File: | 6809-52-5.mol | 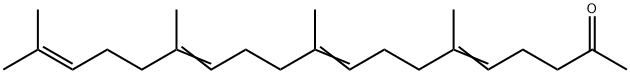 |
| | Teprenone Chemical Properties |
| Boiling point | bp0.01 155-160° | | density | d420.5 0.9081 | | refractive index | nD20 1.4947 | | storage temp. | -20°C | | solubility | DMSO: >5mg/mL | | form | liquid | | color | clear | | Merck | 14,9228 | | Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20° for up to 1 month. | | Cosmetics Ingredients Functions | SKIN CONDITIONING | | InChI | InChI=1S/C23H38O/c1-19(2)11-7-12-20(3)13-8-14-21(4)15-9-16-22(5)17-10-18-23(6)24/h11,13,15,17H,7-10,12,14,16,18H2,1-6H3 | | InChIKey | SBOCYPUKNYDTSX-DMLVRTFPSA-N | | SMILES | CC(=O)CCC=C(C)CCC=C(C)CCC=C(C)CC/C=C(/C)\C | | LogP | 8.200 (est) | | CAS DataBase Reference | 6809-52-5(CAS DataBase Reference) |
| WGK Germany | 3 | | RTECS | RA5391300 | | HS Code | 2914.19.0000 | | Storage Class | 10 - Combustible liquids |
| | Teprenone Usage And Synthesis |
| Description | Teprenone (geranylgeranylacetone) is an antiulcer agent whose synthesis was
inspired by the gastric protecting properties of Coenzyme Q10. Teprenone is
reported to act via enhancement of gastric mucosal glycoprotein secretion. | | Description | Geranylgeranylacetone is an inducer of heat shock protein (Hsp) expression that has been shown to increase Hsp70 (also known as Hsp72 and HspA1A), Hsp22 (HspB8), Hsp27 (HspB1), Hsp90 (HspC), and Hsp105 (HspH1) levels in various cells and tissues. It is orally bioavailable and has diverse effects in vivo, including hepatoprotective, neuroprotective, and antiulcerative effects. The effects of geranylgeranylacetone on Hsp expression are more pronounced under stress conditions. | | Chemical Properties | Yellow Oil | | Originator | Eisai (Japan) | | Uses | An anti-ulcerative. Geranylgeranylacetone can induce expression of HSP70, HSPB8, and HSPB1. Induction of HSP70 expression is protective against the development of various diseases, such as inflammatory bowel disease, hypoxic/ischemic brain injury and spinal and bulbar muscular atrophy (cytoprotective and anti-inflammatory effects). Reports indicate that GGA protects against NSAID-induced gastric and intestinal lesions by induction of HSP70 expression. Other studies have shown that GGA induces expression of HSPB8 and HSPB1 and reduces the formation of amyloid oligomers as well as insoluble aggregates in HSPB5 R120G TG mice. | | Uses | Geranylgeranylacetone has been used as an inducer of heat shock protein 70 (HSP70) to analyze its protective effects against cerebral ischemia/reperfusion (I/R). | | Definition | ChEBI: A terpene ketone in which a (9E,13E)-geranylgernayl group is bonded to one of the alpha-methyls of acetone (it is a mixture of 5E- and 5Z-geoisomers in a 3:2 ratio). | | Brand name | SELBEX | | Biochem/physiol Actions | Geranylgeranylacetone can induce expression of HSP70, HSPB8, and HSPB1. Induction of HSP70 expression is protective against the development of various diseases, such as inflammatory bowel disease, hypoxic/ischemic brain injury and spinal and bulbar muscular atrophy (cytoprotective and anti-inflammatory effects). Reports indicate that GGA protects against NSAID-induced gastric and intestinal lesions by induction of HSP70 expression. Other studies have shown that GGA induces expression of HSPB8 and HSPB1 and reduces the formation of amyloid oligomers as well as insoluble aggregates in HSPB5 R120G TG mice. | | Synthesis | The 3-racemic-keto ester 11 (0.07 g, 0.174 mmol) was used as a raw material, which was mixed with MeOH (0.3 mL) and 5N KOH aqueous solution (0.15 mL), and heated to react at 80-85 °C for 2 h. The progress of the reaction was monitored by TLC. Upon completion of the reaction, the reaction mixture was cooled and acidified with 2N HCl, followed by extraction with ether, ethyl acetate or hexane (3 x 400 mL). The organic layers were combined and washed sequentially with water, aqueous NaHCO3 and brine, and then dried with anhydrous MgSO4. After removing the solvent under reduced pressure, the oily crude product obtained was purified by silica gel column chromatography with a gradient elution of hexane to 1-2% EtOAc in hexane solution as eluent to end up with the colorless liquid 5-trans-GGA 12. Yield: 0.028 g (50%). TLC Rf value: 0.45 (5% EtOAc/hexane); LCMS data: MS (m/z): 333 (MH+); 353 (M+Na). | | References | [1] MANAL M. KAMAL Ola M O. The role of heat shock protein 70 induced by geranylgeranylacetone in carbon tetrachloride-exposed adult rat testes[J]. Pathophysiology, 2013, 20 2: Pages 139-146. DOI:10.1016/j.pathophys.2013.03.003
[2] ANTON LENNIKOV. Induction of heat shock protein 70 ameliorates ultraviolet-induced photokeratitis in mice.[J]. International Journal of Molecular Sciences, 2013, 14 1: 2175-2189. DOI:10.3390/ijms14012175
[3] HIRONORI ADACHI. An acylic polyisoprenoid derivative, geranylgeranylacetone protects against visceral adiposity and insulin resistance in high-fat-fed mice.[J]. American journal of physiology. Endocrinology and metabolism, 2010: E764-71. DOI:10.1152/ajpendo.00075.2010
[4] TETSURO MARUNOUCHI . Protective effect of geranylgeranylacetone via enhanced induction of HSPB1 and HSPB8 in mitochondria of the failing heart following myocardial infarction in rats[J]. European journal of pharmacology, 2014, 730: Pages 140-147. DOI:10.1016/j.ejphar.2014.02.037 |
| | Teprenone Preparation Products And Raw materials |
| Raw materials | BFYVSGCRUDETNO-JYQWTDEISA-N-->Butanoic acid, 3-oxo-, (4E,8E)-1-ethenyl-1,5,9,13-tetramethyl-4,8,12-tetradecatrien-1-yl ester-->GERANYL GERANYL BROMIDE-->ethyl (4E,8E,12E)-2-acetyl-5,9,13,17-tetramethyloctadeca-4,8,12,16-tetraenoate-->GERANYLGERANIOL-->Geranyl linalool-->5-ACETYL-2,2-DIMETHYL-1,3-DIOXANE-4,6-DIONE-->Potassium hydroxide-->Methanol-->Water |
|