|
|
| | 5-Hydroxy-3-methylpyridine-2-carbonitrile Basic information |
| Product Name: | 5-Hydroxy-3-methylpyridine-2-carbonitrile | | Synonyms: | 2-Carbonitrile-5-Hydroxy-3-methylpyridine;5-hydroxy-3-Methyl-2-Pyridinecarbonitrile;5-h;5-Hydroxy-3-methylpyridine-2-carbonitrile;2-Pyridinecarbonitrile,5-hydroxy-3-methyl-(9CI);5-HYDROXY-3-METHYLPYRIDINE-2-CARBONITRILE,PURITY:95% MIN(HPLC);2-Pyridinecarbonitrile, 5-hydroxy-3-methyl-;5-hydroxy-3-methylpicolinonitrile | | CAS: | 228867-86-5 | | MF: | C7H6N2O | | MW: | 134.14 | | EINECS: | | | Product Categories: | PYRIDINE | | Mol File: | 228867-86-5.mol | 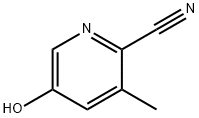 |
| | 5-Hydroxy-3-methylpyridine-2-carbonitrile Chemical Properties |
| Boiling point | 419.7±45.0 °C(Predicted) | | density | 1.26±0.1 g/cm3(Predicted) | | storage temp. | 2-8°C | | pka | 3.36±0.23(Predicted) | | Appearance | Light yellow to yellow Solid | | CAS DataBase Reference | 228867-86-5(CAS DataBase Reference) |
| | 5-Hydroxy-3-methylpyridine-2-carbonitrile Usage And Synthesis |
| Synthesis | Using 5-amino-3-methylpyridine-2-carbonitrile (18.0 g, 135 mmol) as raw material, a 1.6 M aqueous solution of sodium nitrite (10.3 g, 149 mmol) was slowly added to a mixture of its water (243 mL) and concentrated sulfuric acid (67.5 mL) at 0 °C. The reaction mixture was gradually warmed to room temperature and subsequently stirred at 100 °C for 3 hours. Upon completion of the reaction, it was cooled to room temperature and extracted with ethyl acetate (3 x 75 mL). The organic layers were combined, washed sequentially with water (2 × 75 mL) and saturated aqueous sodium chloride solution (2 × 75 mL), dried, filtered and concentrated under reduced pressure to give 5-hydroxy-3-methylpyridine-2-carbonitrile (16 g, 120 mmol, 89% yield) as a yellow solid.1H NMR (400 MHz, DMSO-d6) δ 11.07 (brs, 1H) 8.08 (d, J = 2.6Hz, 1H), 7.20 (d, J = 2.3Hz, 1H), 2.40 (s, 3H). | | References | [1] Patent: WO2015/155626, 2015, A1. Location in patent: Page/Page column 89; 90
[2] Patent: WO2017/51294, 2017, A1. Location in patent: Page/Page column 57; 58
[3] Patent: WO2017/51303, 2017, A1. Location in patent: Page/Page column 72; 73
[4] Journal of Medicinal Chemistry, 2018, vol. 61, # 10, p. 4476 - 4504
[5] Patent: WO2017/51276, 2017, A1. Location in patent: Page/Page column 99; 10016 g |
| | 5-Hydroxy-3-methylpyridine-2-carbonitrile Preparation Products And Raw materials |
|