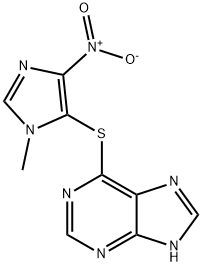
아자티오프린
|
|
아자티오프린 속성
- 녹는점
- 243-244°C
- 끓는 점
- 521.0±60.0 °C(Predicted)
- 밀도
- 1.5379 (rough estimate)
- 굴절률
- 1.7400 (estimate)
- 저장 조건
- 2-8°C
- 용해도
- 디클로로메탄과 디메틸설폭사이드에 용해됩니다.
- 산도 계수 (pKa)
- 8.2(at 25℃)
- 색상
- Me2CO(aq)에서 결정화된 담황색 결정
- 수용성
- 23&C에서 <0.1g/100mL
- Merck
- 14,902
- BCS Class
- 4
- 안정성
- 안정적인. 강한 산화제, 강한 염기와 호환되지 않습니다.
- InChIKey
- LMEKQMALGUDUQG-UHFFFAOYSA-N
- CAS 데이터베이스
- 446-86-6(CAS DataBase Reference)
- IARC
- 1 (Vol. 26, Sup 7, 100A) 2012
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi,T,Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 45-22-36/37/38-20/21/22 | ||
| 안전지침서 | 53-22-26-36/37-45-36 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | UO8925000 | ||
| 위험 등급 | IRRITANT | ||
| HS 번호 | 29339900 | ||
| 유해 물질 데이터 | 446-86-6(Hazardous Substances Data) | ||
| 독성 | LD50 orl-rat: 535 mg/kg NIIRDN 6,3,82 | ||
| 기존화학 물질 | KE-24461 | ||
| 중점관리물질 필터링 | 별표2-56 | ||
| 암, 돌연변이성물질 필터링 | 43 |
아자티오프린 C화학적 특성, 용도, 생산
개요
This immunosuppressive and antineoplastic drug is derived from 6-mercaptopurine. It caused occupational dermatitis in a pharmaceutical worker, reconditioner of old tablet packaging machines and in a production mechanic, working in packaging for a pharmaceutical company.화학적 성질
Azathioprine is a complex heterocyclic compound which forms pale yellow crystals.용도
An immunosuppressive antimetabolite. Also active as disease modifying antirheumatic drug (DMARD). Azathioprine is a purine analog with immunosuppressive effects.정의
ChEBI: A thiopurine that is 6-mercaptopurine in which the mercapto hydrogen is replaced by a 1-methyl-4-nitroimidazol-5-yl group. It is a prodrug for mercaptopurine and is used as an immunosuppressant, prescribed for the treatment of inflammatory conditions and a ter organ transplantation and also for treatment of Crohn's didease and MS.Indications
Azathioprine (Imuran) is a cytotoxic agent that preferentially destroys any rapidly dividing cell. Since immunologically competent cells are generally rapidly dividing cells, azathioprine is very effective as an immunosuppressive drug. Unfortunately, any cell that is replicating is a target for this action. This lack of specificity leads to serious side effects. Azathioprine, in combination with corticosteroids, has historically been used more widely than any other drug in immunosuppressive therapy. It is classified as a purine antimetabolite and is a derivative of 6-mercaptopurine.일반 설명
Pale yellow crystals or yellowish powder. Decomposes at 243-244°C. Used for the treatment of rheumatoid arthritis. A known carcinogen.공기와 물의 반응
Sensitive to oxidation in the air. Insoluble in water.반응 프로필
Azathioprine may react exothermically with acids. Incompatible with isocyanates, peroxides, phenols, epoxides, anhydrides, and acid halides. Hydrolyzed by strongly basic solutions . May react with strong reducing agents to generate flammable gaseous hydrogen or hydrogen sulfide.위험도
Confirmed carcinogen.화재위험
Flash point data for Azathioprine are not available. Azathioprine is probably combustible.색상 색인 번호
This immunosuppressive and antineoplastic drug is derived from 6-mercaptopurine. It caused allergic contact dermatitis in a mother crushing tablets for her leukemic son, and occupational dermatitis in a pharmaceutical reconditioner of old tablet packaging machines, and in a production mechanic working in packaging for a pharmaceutical company.Mechanism of action
Azathioprine is a phase-specific drug that is toxic to cells during nucleic acid synthesis. Phase-specific drugs are toxic during a specific phase of the mitotic cycle, usually the S-phase, when DNA synthesis is occurring, as opposed to cycle-specific drugs that kill both cycling and intermitotic cells.Azathioprine is converted in vivo to thioinosinic acid, which competitively inhibits the synthesis of inosinic acid, the precursor to adenylic acid and guanylic acid. In this way, azathioprine inhibits DNA synthesis and therefore suppresses lymphocyte proliferation.This effectively inhibits both humoral and cell-mediated immune responses.
Pharmacology
Azathioprine is well absorbed following oral administration, with peak blood levels occurring within 1 to 2 hours. It is rapidly and extensively metabolized to 6- mercaptopurine, which is further converted in the liver and erythrocytes to a variety of metabolites, including 6- thiouric acid. Metabolites are excreted in the urine.The half-life of azathioprine and its metabolites in the blood is about 5 hours.Clinical Use
Azathioprine is a relatively powerful antiinflammatory agent. Although its beneficial effect in various conditions is principally attributable to its direct immunosuppressive action, the antiinflammatory properties of the drug play an important role in its overall therapeutic effectiveness.Azathioprine has been used widely in combination with corticosteroids to inhibit rejection of organ transplants, particularly kidney and liver allografts. However, it is usually reserved for patients who do not respond to cyclosporine plus corticosteroids alone.
Azathioprine also has applications in certain disorders with autoimmune components, most commonly rheumatoid arthritis. It is as effective as cyclophosphamide in the treatment of Wegener’s granulomatosis. It has largely been replaced by cyclosporine in immunosuppressive therapy. Relative to other cytotoxic agents, the better oral absorption of azathioprine is the reason for its more widespread clinical use.
부작용
The therapeutic use of azathioprine has been limited by the number and severity of adverse effects associated with its administration. Bone marrow suppression resulting in leukopenia, thrombocytopenia, or both may occur. GI toxicity may be a problem. It is also mildly hepatotoxic. Because of its immunosuppressive activity, azathioprine therapy can lead to serious infections. It has been shown to be mutagenic in animals and humans and carcinogenic in animals.Safety Profile
Confirmed human carcinogen producing bladder tumors and leukemia. Poison by subcutaneous, intradermal, and intraperitoneal routes. Moderately toxic by ingestion. Human systemic effects: liver changes, hypermotility, diarrhea, nausea or vomiting, increased body temperature, BP lowering, decreased urine volume or anuria, normocytic anemia, bone marrow changes. An experimental teratogen. Other experimental reproductive effects. Human mutation data reported. When heated to decomposition it emits very toxic fumes of NO,xand SOx. An immunosuppressant.잠재적 노출
Azathioprine is an immunosuppressive agent, generally used in combination with a corticosteroid to prevent rejection following renal homotransplantations. It also is used following transplantation of other organs. Other uses of azathioprine include the treatment of a variety of presumed autoimmune diseases, including rheumatoid arthritis; ankylosing spondylitis; systemic lupus erythematosus; dermatonyositis, periarteritis nodosa, scleroderma, refractory thombocytopenic purpura; autoimmune hemolytic anemia; chronic active liver disease; regional enteritis; ulcerative colitis; various autoimmune diseases of the eye; acute and chronic glomerulonephritis; the nephritic syndrome; Wegener’s granulomatosis; and multiple sclerosis.Carcinogenicity
Azathioprine is known to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in humans.운송 방법
UN2811 Toxic solids, organic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required.비 호환성
Incompatible with reducing agents, such as hydrides (may cause the release of explosive gases), oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong acids (violent exothermic reaction), strong bases.아자티오프린 준비 용품 및 원자재
원자재
준비 용품
아자티오프린 공급 업체
글로벌( 454)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618740459177 |
sarah@tnjone.com | China | 1142 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21687 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9340 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 |
sales@sdzschem.com | China | 2931 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49391 | 58 |