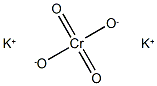
크롬산 칼륨 C화학적 특성, 용도, 생산
화학적 성질
lemon-yellow crystals
용도
Potassium chromate (K2CrO4) is soluble in water and is used to make bright yellow inks
and paint pigments. It is also used as a reagent in chemical laboratories and as a mordant to
“fix” dyes in colored textiles.
정의
ChEBI: A potassium salt consisting of potassium and chromate ions in a 2:1 ratio.
일반 설명
Potassium chromate is a yellow crystalline solid. Potassium chromate is soluble in water. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Potassium chromate is used in chemical analysis, in making pigments for paints and inks, as a fungicide, and to make other chromium compounds.
공기와 물의 반응
Soluble in water.
반응 프로필
Oxidizing agents, such as Potassium chromate , can react with reducing agents to generate heat and products that may be gaseous (causing pressurization of closed containers). The products may themselves be capable of further reactions (such as combustion in the air). The chemical reduction of materials in this group can be rapid or even explosive, but often requires initiation (heat, spark, catalyst, addition of a solvent). Explosive mixtures of inorganic oxidizing agents with reducing agents often persist unchanged for long periods if initiation is prevented. Such systems are typically mixtures of solids, but may involve any combination of physical states. Some inorganic oxidizing agents are salts of metals that are soluble in water; dissolution dilutes but does not nullify the oxidizing power of such materials. Organic compounds, in general, have some reducing power and can in principle react with compounds in this class. Actual reactivity varies greatly with the identity of the organic compound. Inorganic oxidizing agents can react violently with active metals, cyanides, esters, and thiocyanates.
위험도
Toxic by ingestion and inhalation.
건강위험
Inhalation causes local irritation of mucous membranes; continuing nose irritation can result in perforation of nasal septum. Ingestion may cause violent gastroenteritis, circulatory collapse, vertigo, coma, and toxic nephritis; ingestion of excessive quantities can be fatal. Contact with eyes causes severe irritation and conjunctivitis. Repeated or prolonged exposure to dust, mist, or solutions may cause dermatitis; contact with breaks in the skin may cause ``chrome sores'' appearing as slow-healing, hard-rimmed ulcers which leave the area vulnerable to infection.
화재위험
Behavior in Fire: May increase intensity of fire if in contact with combustible materials. Cool containers and spilled material with plenty of water.
Safety Profile
Confirmed carcinogen
with experimental tumorigenic data. Poison
by ingestion, intravenous, subcutaneous, and
intramuscular routes. An experimental
teratogen. Other experimental reproductive
effects. Human mutation data reported. A
powerful oxidizer. When heated to
decomposition it emits toxic fumes of K2O.
Used as a mordant for wool, in the oxidizing
and treatment of dyes on materials. See also
CHROMIUM COMPOUNDS.
잠재적 노출
Potassium chromate is used in printing: photomechanical processing; chrome-pigment production; and wool preservative methods; to make dyes, pigments, inks and enamels; as an oxidizing agent; analytical reagent; in electroplating; explosives.
운송 방법
UN1479 Oxidizing solid, n.o.s., Hazard Class: 5.1; Labels: 5.1-Oxidizer, Technical Name Required. UN3288 Toxic solids, inorganic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required
Purification Methods
Crystallise it from conductivity water (0.6g/mL at 20o), and dry it between 135o and 170o.
비 호환성
A powerful oxidizer. Violent reactions with combustibles, organics, powdered metals; or easily oxidizable substances. Contact with hydroxylamine, hydrazine causes explosion.
크롬산 칼륨 준비 용품 및 원자재
원자재
준비 용품