Selenious acid suppliers
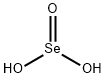
Selenious acid
- CAS:
- 7783-00-8
- MF:
- H2O3Se
- MW:
- 128.97
Properties
- Melting point:
- 70 °C (dec.) (lit.)
- Boiling point:
- 684.9 °C(lit.)
- Density
- 3.004 g/mL at 25 °C (lit.)
- vapor pressure
- 2.7hPa at 15℃
- Flash point:
- 690°C
- storage temp.
- Store at RT.
- solubility
- H2O: soluble
- form
- powder
- pka
- 2.46(at 25℃)
- color
- White to almost white or light pink
- Specific Gravity
- 3.004
- PH
- 3.15(1 mM solution);2.47(10 mM solution);1.9(100 mM solution)
- Water Solubility
- 167 g/100 mL (20 ºC)
- Sensitive
- Hygroscopic
- Merck
- 14,8430
- Exposure limits
- ACGIH: TWA 0.2 mg/m3
NIOSH: IDLH 1 mg/m3; TWA 0.2 mg/m3
- Stability:
- Stable.
Safety Information
- Symbol(GHS)



GHS06,GHS08,GHS09
- Signal word
- Danger
- Hazard statements
- H301+H331-H373-H410
- Precautionary statements
- P273-P301+P310+P330-P304+P340+P311-P314
- Hazard Codes
- T,N,Xi
- Risk Statements
- 36/38-50/53-33-23/25-52/53
- Safety Statements
- 61-60-45-28A-20/21-26-28
- RIDADR
- UN 3440 6.1/PG 3
- WGK Germany
- 3
- RTECS
- VS7700000
- F
- 3
- TSCA
- Yes
- HazardClass
- 8
- PackingGroup
- III
- HS Code
- 28111980
Use
Selenious acid is a colourless, deliquescent crystal in appearance. It decomposes on heating, producing water and toxic fumes of selenium oxides. It is incompatible with strong reducing agents, organic materials, and finely powdered metals and reacts on contact with acids producing toxic gaseous hydrogen selenide. Selenious acid is a non-combustible chemical. On fire/burning, it emits irritating or toxic fumes (or gases). The major use of selenious acid is in changing the colour of steel, especially the steel in guns from silver grey to blue grey. It is also used for the chemical darkening and patination of copper brass and bronze, producing a rich dark-brown colour that can be further enhanced with mechanical abrasion.
According to relevant laws, regulations, policies and the provisions of this website, this website does not display the product information!
The source is "剧毒、有毒、易致癌化学品". If the information is inaccurate, please contact the platform to modify it.
contact information:info@chemicalbook.com 400-158-660