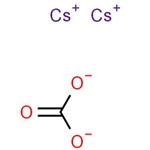
- Caesium carbonate
-
- $4.00 / 25g
-
2024-02-23
- CAS:534-17-8
- Min. Order: 25g
- Purity: 0.99
- Supply Ability: 25kg
- Caesium fluoride
-

- $20.00 / 1kg
-
2023-02-18
- CAS:13400-13-0
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 5000 Ton
- Caesium fluoride
-

- $15.00 / 1KG
-
2021-07-02
- CAS:13400-13-0
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
|
| | Caesium Bromide Basic information |
| | Caesium Bromide Chemical Properties |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36 | | WGK Germany | 3 | | RTECS | FK9275000 | | TSCA | Yes | | HS Code | 28275900 | | Toxicity | LD50 i.p. in rats: 1.4 g/kg (Cochran) |
| | Caesium Bromide Usage And Synthesis |
| Physical properties |
Caesium bromide is an ionic compound of caesium and bromine. It is a white or transparent solid with melting point at 636 °C that readily dissolves in water.

| | Uses | Cesium bromide is used as reagent, intermediate in pharmaceuticals.Cesium bromide crystals are used in scintillation counters to detect radiation. The compound is also used to coat the inside of fluorescent screens.
| | Safety | Moderately toxic by intraperitoneal route.When heated to decomposition it emits toxic fumes of Br-. | | Synthesis | Caesium bromide can be prepared via following reactions:
Neutralization:
CsOH (aq) + HBr (aq) → CsBr (aq) + H2O (l)
Cs2(CO3) (aq) + 2 HBr (aq) → 2 CsBr (aq) + H2O (l) + CO2(g)
| | Metabolism | Bromide is mainly absorbed via inhalation, but may also enter the body through dermal contact. Bromine salts can be ingested. Due to its reactivity, bromine quickly forms bromide and may be deposited in the tissues, displacing other halogens. |
| | Caesium Bromide Preparation Products And Raw materials |
|