|
|
| | UNCARINE E Basic information |
| Product Name: | UNCARINE E | | Synonyms: | allo-Isopteropodine;UNCARINE E;(7alpha,19alpha,20alpha)-19-Methyl-2-oxoformosanan-16-carboxylic acid methyl ester;ISOPTEROPODINE;(7S,20α)-19α-Methyl-2-oxoformosanan-16-carboxylic acid methyl ester;7-Isopteropodine;(1R,4aR,10aR)-2'-keto-1-methyl-spiro[1,4a,5,5a,7,8,10,10a-octahydropyrano[3,4-f]indolizine-6,3'-indoline]-4-carboxylic acid methyl ester;methyl (1R,4aR,10aR)-1-methyl-2'-oxo-spiro[1,4a,5,5a,7,8,10,10a-octahydropyrano[3,4-f]indolizine-6,3'-1H-indole]-4-carboxylate | | CAS: | 5171-37-9 | | MF: | C21H24N2O4 | | MW: | 368.43 | | EINECS: | | | Product Categories: | standard material,plant extracts | | Mol File: | 5171-37-9.mol | 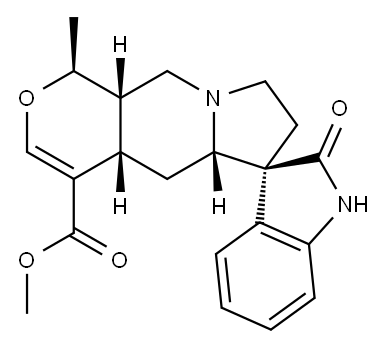 |
| | UNCARINE E Chemical Properties |
| Melting point | 209-211 °C | | Boiling point | 555.2±50.0 °C(Predicted) | | density | 1.33 | | storage temp. | -20°C Freezer | | solubility | Chloroform (Slightly), Methanol (Slightly) | | pka | 13.56±0.60(Predicted) | | form | Solid | | color | Pale Yellow to Pale Beige |
| | UNCARINE E Usage And Synthesis |
| Description | This alkaloid occurs in several species of Uncaria and yields colourless crystals
from MeOH. It is laevorotatory with [α]D - 96° (CHCI3). The structure has
been determined by spectroscopic methods. | | Uses | Isopteropodine is an alkaloid extract from Uncaria Tomentosathat is believed to possess antiproliferative and pro-apoptotic effects. | | Definition | ChEBI: Uncarine E is a member of indolizines. | | References | Hart, Johns, Lamberton., Chem. Commun., 87 (1967)
Hemingway, Phillipson., 1. Pharm. Pharmacol., 24, 169P (1972) |
| | UNCARINE E Preparation Products And Raw materials |
|