- Dehydronandrolon
-

- $60.00 / 1kg
-
2024-05-23
- CAS:2590-41-2
- Min. Order: 1kg
- Purity: 99.99%
- Supply Ability: 1000
- Dehydronandrolon
-

- $11.00 / 1kg
-
2024-05-11
- CAS:2590-41-2
- Min. Order: 0.10000000149011612kg
- Purity: 99%
- Supply Ability: 10 tons
- Dehydronandrolon
-

- $1.00 / 1g
-
2024-05-11
- CAS:2590-41-2
- Min. Order: 1000g
- Purity: 98%
- Supply Ability: 20ton
|
| | Dehydronandrolone Acetate Basic information |
| Product Name: | Dehydronandrolone Acetate | | Synonyms: | 17-ACETOXYESTRA-4,6-DIEN-3-ONE;DEHYDRONANDROLON;(8R,9S,13S,14S,17S)-13-Methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate;Dehydronandrolone-6 Acetate;Acetic acid 13-Methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl este;(8R,9S,10R,13S,14S,17S)-13-Methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate;Estra-4,6-dien-3-one,17-(acetyloxy)-, (17b)-;Dehydronandrolon 17b-Acetyloxy-estra-4,6-diene-3-one | | CAS: | 2590-41-2 | | MF: | C20H26O3 | | MW: | 314.42 | | EINECS: | 643-000-1 | | Product Categories: | API;Intermediates;Intermediates & Fine Chemicals;Pharmaceuticals;Steroids | | Mol File: | 2590-41-2.mol | 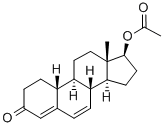 |
| | Dehydronandrolone Acetate Chemical Properties |
| Melting point | 95-98?C | | Boiling point | 457.4±45.0 °C(Predicted) | | density | 1.15±0.1 g/cm3(Predicted) | | storage temp. | Controlled Substance, -20°C Freezer | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Solid | | color | Off-White to Pale Yellow | | InChI | InChI=1/C20H26O3/c1-12(21)23-19-8-7-18-17-5-3-13-11-14(22)4-6-15(13)16(17)9-10-20(18,19)2/h3,5,11,15-19H,4,6-10H2,1-2H3/t15-,16+,17+,18-,19-,20-/s3 | | InChIKey | OGUASZAAVFYYIL-SBVYDUMINA-N | | SMILES | [C@@]12([H])CC[C@H](OC(=O)C)[C@@]1(C)CC[C@]1([H])[C@@]3([H])CCC(=O)C=C3C=C[C@@]21[H] |&1:0,4,9,13,15,25,r| | | CAS DataBase Reference | 2590-41-2(CAS DataBase Reference) |
| | Dehydronandrolone Acetate Usage And Synthesis |
| Description |
Dehydronandrolone acetate is a critical precursor in the synthesis of C7-functionalized steroidal drugs such as the new anti-breast cancer drug Fulvestrant, the treatment of menopause and postmenopausal syndrome drug Tibolone, as well as the highly effective protein anabolic hormone Mibolone. Nandrolone as starting material was first subjected to simultaneous acylation with acetic anhydride and acetyl chloride to form the intermediate, which was then brominated with N-bromosuccinimide (NBS) under basic conditions to eliminate one molecule of HBr to produce Dehydronandrolone acetate (73% yield)[1].

| | Chemical Properties | Pale Yellow Solid | | Uses | Tibolone intermediate. | | References | [1] Zili Zhang and Aitao Li*. “A Designed Chemoenzymatic Route for Efficient Synthesis of 6-Dehydronandrolone Acetate: A Key Precursor in the Synthesis of C7-Functionalized Steroidal Drugs.” ACS Catalysis 13 19 (2023): 13111–13116.
|
| | Dehydronandrolone Acetate Preparation Products And Raw materials |
|