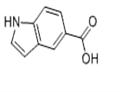
Indole-5-carboxylic acid
- CAS No.
- 1670-81-1
- Chemical Name:
- Indole-5-carboxylic acid
- Synonyms
- 1H-INDOLE-5-CARBOXYLIC ACID;JACS-1670-81-1;5-CARBOXYINDOLE;5-Carboxy-1H-indole;RARECHEM AL BO 0206;TIMTEC-BB SBB003951;Indolecarboxylicacid,5-;5-INDOLECARBOXYLIC ACID;INDOLE-5-CARBOXYLIC AICD;INDOLE-5-CARBOXYLIC ACID
- CBNumber:
- CB0736428
- Molecular Formula:
- C9H7NO2
- Molecular Weight:
- 161.16
- MDL Number:
- MFCD00005678
- MOL File:
- 1670-81-1.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Indole-5-carboxylic Acid >98.0%(GC)(T) | I0029 | 1g | $47 |
| Indole-5-carboxylic Acid >98.0%(GC)(T) | I0029 | 5g | $91 |
| Indole-5-carboxylic Acid | I627005 | 50g | $525 |
| Indole-5-carboxylic Acid | I627005 | 1g | $45 |
| Indole-5-carboxylic Acid | I627005 | 5g | $120 |
| More product size | |||
| Melting point | 211-213 °C (lit.) |
|---|---|
| Boiling point | 287.44°C (rough estimate) |
| Density | 1.2480 (rough estimate) |
| refractive index | 1.5050 (estimate) |
| storage temp. | Keep in dark place,Sealed in dry,Room Temperature |
| solubility | Soluble in ethanol (50 mg/ml), dimethyl sulfoxide and methanol. |
| pka | 4.40±0.30(Predicted) |
| form | Powder |
| color | Light beige to yellow |
| BRN | 124391 |
| InChI | InChI=1S/C9H7NO2/c11-9(12)7-1-2-8-6(5-7)3-4-10-8/h1-5,10H,(H,11,12) |
| InChIKey | IENZCGNHSIMFJE-UHFFFAOYSA-N |
| SMILES | N1C2=C(C=C(C(O)=O)C=C2)C=C1 |
| CAS DataBase Reference | 1670-81-1(CAS DataBase Reference) |
| UNSPSC Code | 12352100 |
| NACRES | NA.22 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P264-P280-P302+P352+P332+P313+P362+P364-P305+P351+P338+P337+P313-P261-P280a-P304+P340-P305+P351+P338-P405-P501a | |||||||||
| PPE | Eyeshields, Gloves, type N95 (US) | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 36/37/38-21/22 | |||||||||
| Safety Statements | 22-24/25-36/37/39-26 | |||||||||
| WGK Germany | 3 | |||||||||
| HazardClass | IRRITANT | |||||||||
| HS Code | 29339990 | |||||||||
| Storage Class | 13 - Non Combustible Solids | |||||||||
| NFPA 704 |
|
Indole-5-carboxylic acid price More Price(31)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| TCI Chemical | I0029 | Indole-5-carboxylic Acid >98.0%(GC)(T) | 1670-81-1 | 1g | $47 | 2026-03-19 | Buy |
| TCI Chemical | I0029 | Indole-5-carboxylic Acid >98.0%(GC)(T) | 1670-81-1 | 5g | $91 | 2026-03-19 | Buy |
| TRC | I627005 | Indole-5-carboxylic Acid | 1670-81-1 | 50g | $525 | 2021-12-16 | Buy |
| TRC | I627005 | Indole-5-carboxylic Acid | 1670-81-1 | 1g | $45 | 2021-12-16 | Buy |
| TRC | I627005 | Indole-5-carboxylic Acid | 1670-81-1 | 5g | $120 | 2021-12-16 | Buy |
Indole-5-carboxylic acid Chemical Properties,Uses,Production
Chemical Properties
light beige to yellow powder
Uses
Reactant for preparation of tryptophan dioxygenase inhibitors pyridyl-ethenyl-indoles, as potential anticancer immunomodulators 1 Reactant for preparation of indolyl-quinolines via metal- and solvent-free autoxidative coupling reaction 2 Reactant for preparation of anthranilic acids using bromamine-B oxidant and palladium chloride catalyst 3 Reactant for synthesis of indirubin derivatives 4 Reactant for preparation of amide conjugates with ketoprofen, as inhibitors of Gli1-mediated transcription in Hedgehog pathway 5 Reactant for preparation of piperazine-bisamide analogs as human growth hormone secretagogue receptor antagonists for treatment of obesity.
Uses
Indole-5-carboxylic acid is the suitable reagent used to study the intermolecular excited state proton transfer in indole-2-carboxylic acid and indole-5-carboxylic acid in various solvents in acidic, basic, and neutral media by steady state and time resolved fluorescence spectroscopy. It may be used in the electrochemical synthesis of poly(indole-5-carboxylic acid) (PICA) films. Also used as reactant for preparation of tryptophan dioxygenase inhibitors pyridyl-ethenyl-indoles, as potential anticancer immunomodulators, synthesis of indirubin derivatives and amide conjugates with ketoprofen, as inhibitors of Gli1-mediated transcription in Hedgehog pathway.
Uses
Indole-5-carboxylic acid is the suitable reagent used to study the intramolecular excited state proton transfer in indole-2-carboxylic acid and indole-5-carboxylic acid in various solvents in acidic, basic, and neutral media by steady state and time resolved fluorescence spectroscopy. It may be used in the electrochemical synthesis of poly(indole-5-carboxylic acid) (PICA) films.
Definition
ChEBI: Indole-5-carboxylic acid is an indolecarboxylic acid in which the carboxy group is the only substituent and is located at position 5. It has a role as a plant metabolite.
General Description
Indole-5-carboxylic acid is an indole derivative. On electropolymerization, it affords electroactive polymer film of poly(indole-5-carboxylic-acid). Different concentrations of indole-5-carboxylic acid in sulfuric acid solution has been investigated for the preventive action against mild steel corrosion. On electropolymerization it affords a trimeric product. Characterization studies of the trimeric product by 1H NMR and various one- and two-dimensional NMR techniques have been reported.
Synthesis
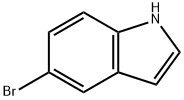
10075-50-0

124-38-9

1670-81-1
To a solution of 5-bromo-1H-indole (0.86 g, 4.4 mmol, 1.0 eq.) in anhydrous THF (20 mL) was slowly added 2 M isopropylmagnesium chloride (i-PrMgCl) in a solution of THF (2.2 mL, 4.4 mmol, 1.0 eq.) at 0 °C, keeping the reaction temperature at 0 °C. After addition, the clarified solution was continued to be stirred at the same temperature for 5 minutes. Subsequently, a hexane solution (3.5 mL, 8.8 mmol, 2.0 eq.) of 2.5 M n-butyllithium (n-BuLi) was added dropwise over 5 min, controlling the reaction temperature to not exceed 20 °C. After addition, the reaction mixture was stirred at 0 °C for 30 min. Then, dry carbon dioxide gas (0.2 g, 4.4 mmol, 1.0 eq.) was passed into the reaction system and the reaction mixture was slowly warmed up to 20°C over 0.5 hr. Upon completion of the reaction, the reaction was quenched with deionized water (6 mL) and stirring was continued for 10 min. The organic and aqueous phases were separated and the aqueous phase was extracted once with ethyl acetate (10 mL). The organic phases were combined, and after the suspension was returned to room temperature, it was filtered through a silica gel pad (0.51 cm, eluting with 10 mL of ethyl acetate). The filtrate was concentrated and the residue was purified by rapid column chromatography on silica gel (eluent: petroleum ether/ethyl acetate = 3:1) to afford indole-5-carboxylic acid (3das) as an off-white solid (0.46 g, yield: 65%), melting point: 210 °C. 1H NMR (600 MHz, DMSO) δ 12.39 (s, 1H), 11.46 (s, 1H), 8.25 (s, 1H), 7.72 (dd, J = 8.5, 1.5 Hz, 1H), 7.45 (dd, J = 8.4, 5.7 Hz, 2H), 6.57 (s, 1H). 13C NMR (151 MHz, DMSO) δ 168.90, 138.80, 127.64, 127.35, 123.28, 122.67, 121.87, 111.57, 102.93.
References
[1] Molecules, 2017, vol. 22, # 11,
[2] Journal of the American Chemical Society, 2017, vol. 139, # 28, p. 9467 - 9470
Indole-5-carboxylic acid Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of2
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 33024 | 60 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29821 | 58 |
| Zhejiang ZETian Fine Chemicals Co. LTD | +8618957127338 | stella@zetchem.com | China | 2994 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 |
| Wuhan Chemwish Technology Co., Ltd | 027-67849912 | sales@chemwish.com | CHINA | 10821 | 58 |
| Nanjing jinheyou Trading Co., Ltd. | 15705188088 | admin@jheyou.com | CHINA | 1268 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| SIMAGCHEM CORP | +86-5922680277 +86-13806087780 | sale@simagchem.com | China | 17346 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34563 | 58 |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 | eric@witopchemical.com | China | 23541 | 58 |
View Lastest Price from Indole-5-carboxylic acid manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2021-08-12 | Indole-5-carboxylic acid
1670-81-1
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd | |
 |
2020-06-02 | Indole-5-carboxylic acid in stock Factory
1670-81-1
|
US $1.00 / KG | 1KG | TOP 3 Factory in China | Top 3 production capacity Factory | Chemwill Asia Co.,Ltd. | |
 |
2019-12-23 | Indole-5-carboxylic acid
1670-81-1
|
US $0.00 / g | 1g | 95%min | 20kg/month | Nanjing jinheyou Trading Co., Ltd. |
-

- Indole-5-carboxylic acid
1670-81-1
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd
-

- Indole-5-carboxylic acid in stock Factory
1670-81-1
- US $1.00 / KG
- TOP 3 Factory in China
- Chemwill Asia Co.,Ltd.
-

- Indole-5-carboxylic acid
1670-81-1
- US $0.00 / g
- 95%min
- Nanjing jinheyou Trading Co., Ltd.





