tert-butyl hydroxy(methyl)carbamate
- CAS No.
- 19689-97-5
- Chemical Name:
- tert-butyl hydroxy(methyl)carbamate
- Synonyms
- N-Boc-N-methylhydroxylamine;tert-butyl hydroxy(methyl)carbamate;tert-?Butyl N-?hydroxy-?N-?methylcarbamate;Carbamic acid, N-hydroxy-N-methyl-,1,1-dimethylethyl ester
- CBNumber:
- CB23098385
- Molecular Formula:
- C6H13NO3
- Molecular Weight:
- 147.17
- MDL Number:
- MFCD24465554
- MOL File:
- 19689-97-5.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| tert-Butylhydroxy(methyl)carbamate | B943435 | 100mg | $60 |
| tert-Butylhydroxy(methyl)carbamate 97% | AK659140 | 1g | $115 |
| tert-Butyl hydroxy(methyl)carbamate 95% | 182265 | 5g | $743 |
| tert-Butylhydroxy(methyl)carbamate 97% | AK659140 | 10g | $821 |
| Tert-butylhydroxy(methyl)carbamate | 7005CB | 10g | $1068 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P264-P280-P302+P352-P321-P332+P313-P362-P264-P280-P305+P351+P338-P337+P313P |
tert-butyl hydroxy(methyl)carbamate price
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| TRC | B943435 | tert-Butylhydroxy(methyl)carbamate | 19689-97-5 | 100mg | $60 | 2021-12-16 | Buy |
| Ark Pharm | AK659140 | tert-Butylhydroxy(methyl)carbamate 97% | 19689-97-5 | 1g | $115 | 2021-12-16 | Buy |
| Matrix Scientific | 182265 | tert-Butyl hydroxy(methyl)carbamate 95% | 19689-97-5 | 5g | $743 | 2021-12-16 | Buy |
| Ark Pharm | AK659140 | tert-Butylhydroxy(methyl)carbamate 97% | 19689-97-5 | 10g | $821 | 2021-12-16 | Buy |
| AK Scientific | 7005CB | Tert-butylhydroxy(methyl)carbamate | 19689-97-5 | 10g | $1068 | 2021-12-16 | Buy |
tert-butyl hydroxy(methyl)carbamate Chemical Properties,Uses,Production
Synthesis
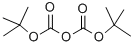
24424-99-5

4229-44-1
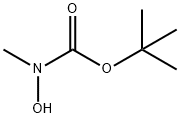
19689-97-5
The general procedure for synthesizing tert-butyl hydroxy(methyl)carbamate from di-tert-butyl dicarbonate and N-methylhydroxylamine hydrochloride is as follows: 1. in a dry reaction flask, dissolve N-methylhydroxylamine hydrochloride in an appropriate amount of anhydrous organic solvent, such as dichloromethane or tetrahydrofuran. 2. An equimolar amount of di-tert-butyl dicarbonate is slowly added while the reaction mixture is stirred under cooling in an ice bath. 3. the reaction mixture was continued to be stirred at room temperature for 4-6 hours and the progress of the reaction was monitored by thin layer chromatography (TLC). 4. Upon completion of the reaction, the reaction was quenched with water and the product was extracted with an organic solvent. 5. The organic phases were combined, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give the crude product. 6. The crude product was purified by column chromatography to obtain the target compound tert-butyl hydroxy(methyl)carbamate. Example 4: Synthesis of NOOTA10107 containing N-methylhydroxylamine According to Scheme 4, the steps for synthesizing N-methylhydroxylamine-containing NOOTA linker are as follows: 1. in accordance with the general steps described above, tert-butyl hydroxy(methyl)carbamate was synthesized from di-tert-butyl dicarbonate and N-methylhydroxylamine hydrochloride. 2. the obtained tert-butyl hydroxy(methyl)carbamate is further reacted with the precursor compound of NOTA linker, and the specific reaction conditions and steps are to be adjusted according to the structure and requirements of NOTA linker. 3. After completion of the reaction, the NOTA linker containing N-methylhydroxylamine is purified by an appropriate purification method, such as column chromatography or recrystallization.
References
[1] Journal of the American Chemical Society, 2013, vol. 135, # 31, p. 11521 - 11524
[2] Organic Letters, 2005, vol. 7, # 25, p. 5729 - 5732
[3] Organic Letters, 2007, vol. 9, # 20, p. 4009 - 4012
[4] Chemical & Pharmaceutical Bulletin, 1982, vol. 30, # 4, p. 1221 - 1224
[5] Organic Letters, 2009, vol. 11, # 22, p. 5210 - 5213

tert-butyl hydroxy(methyl)carbamate Preparation Products And Raw materials
tert-butyl hydroxy(methyl)carbamate Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| ShenZhen Trendseen Biological Technology Co.,Ltd. | 13417589054 | trendseenbio@gmail.com | China | 11681 | 58 |
| Aladdin Scientific | tp@aladdinsci.com | United States | 52923 | 58 | |
| Amadis Chemical Company Limited | 571-89925085 | sales@amadischem.com | China | 131957 | 58 |
| Shanghai Raise Chemical Technology Co.,Ltd | 15026594951 | rs@raise-chem.com | China | 4677 | 55 |
| Mashilabs (Shanghai) Co.,Ltd. | 19916721580 | mafarm@126.com | China | 4539 | 58 |
| Struchem Co., Ltd. | 0512-63009836 18994340901 | helen@struchem.com | China | 3979 | 60 |
| Henan Alpha Chemical Co., Ltd. | 0371-55055611 18137792234 | 3957071458@qq.com | China | 9908 | 58 |
| Energy Chemical | 021-58432009 400-005-6266 | marketing1@energy-chemical.com | China | 44896 | 58 |
| Nanjing Shizhou Biology Technology Co.,Ltd | 13675144456 | sean.lv@synzest.com | China | 10784 | 58 |
| Hangzhou Taorui Biotechnology Co., Ltd | 15268198094 | sales@torreybio.com | China | 3952 | 58 |
19689-97-5(tert-butyl hydroxy(methyl)carbamate)Related Search:
1of3





