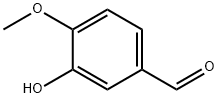
Glycitein
- CAS No.
- 40957-83-3
- Chemical Name:
- Glycitein
- Synonyms
- Gycitein;GLYCITEIN;GLYCETEIN; Glicetein;Glycitein-RM;Glycitein(SH);Glycitein, >=98%;Glycitein (15 mg);Glycitein USP/EP/BP;GLYCITEIN,FROMSOYBEAN
- CBNumber:
- CB7253674
- Molecular Formula:
- C16H12O5
- Molecular Weight:
- 284.26
- MDL Number:
- MFCD00016679
- MOL File:
- 40957-83-3.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Glycitein phyproof? Reference Substance | PHL89692 | 10mg | $361 |
| Glycitein analytical standard | 43534 | 10mg | $350 |
| Glycitein United States Pharmacopeia (USP) Reference Standard | 1295844 | 15mg | $1260 |
| Glycitein | G0634 | 25MG | $182 |
| Glycitein ≥98% | 14162 | 5mg | $49 |
| More product size | |||
| Melting point | >300°C |
|---|---|
| Boiling point | 547.4±50.0 °C(Predicted) |
| Density | 1.420±0.06 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | Aqueous Base (Slightly), DMSO (Slightly) |
| pka | 7.03±0.20(Predicted) |
| form | Solid |
| color | Light Brown |
| biological source | synthetic (organic) |
| InChI | InChI=1S/C16H12O5/c1-20-15-6-11-14(7-13(15)18)21-8-12(16(11)19)9-2-4-10(17)5-3-9/h2-8,17-18H,1H3 |
| InChIKey | DXYUAIFZCFRPTH-UHFFFAOYSA-N |
| SMILES | C1OC2=CC(O)=C(OC)C=C2C(=O)C=1C1=CC=C(O)C=C1 |
| LogP | 2.570 (est) |
| CAS DataBase Reference | 40957-83-3(CAS DataBase Reference) |
| EWG's Food Scores | 1 |
| FDA UNII | 92M5F28TVF |
| UNSPSC Code | 85151701 |
| NACRES | NA.51 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS06 |
|---|---|
| Signal word | Danger |
| Hazard statements | H301-H311-H314-H331 |
| Precautionary statements | P260-P264-P270-P271-P280-P301+P330+P331-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P310-P403+P233-P405-P501 |
| PPE | Eyeshields, Gloves, type N95 (US) |
| Hazard Codes | Xi |
| Safety Statements | 24/25 |
| WGK Germany | 3 |
| HS Code | 29329990 |
| Storage Class | 11 - Combustible Solids |
Glycitein price More Price(41)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | PHL89692 | Glycitein phyproof? Reference Substance | 40957-83-3 | 10mg | $361 | 2026-03-19 | Buy |
| Sigma-Aldrich | 43534 | Glycitein analytical standard | 40957-83-3 | 10mg | $350 | 2026-03-19 | Buy |
| Sigma-Aldrich | 1295844 | Glycitein United States Pharmacopeia (USP) Reference Standard | 40957-83-3 | 15mg | $1260 | 2026-03-19 | Buy |
| TCI Chemical | G0634 | Glycitein | 40957-83-3 | 25MG | $182 | 2026-03-19 | Buy |
| Cayman Chemical | 14162 | Glycitein ≥98% | 40957-83-3 | 5mg | $49 | 2024-03-01 | Buy |
Glycitein Chemical Properties,Uses,Production
Description
Glycitein is an O-
Chemical Properties
Pale Orange Solid
Uses
Glycitein may be used as a reference standard in the determination of glycitein in hydrolyzed dry soya extracts using reversed-phase high-performance liquid chromatography (RP-HPLC).
Uses
Glycitein has been used as standard for the analysis of soy isoflavones and metabolites in urine. It has also been used to bind to recombinant estrogen and progesterone receptors to know the relative binding affinity (RBA) for detection of potential endocrine disruptors.
Uses
The compund shows an anti-cancer activity, plasma cholesterol reduction, reduction in postmenopausal bone loss
Definition
ChEBI: A methoxyisoflavone that is isoflavone substituted by a methoxy group at position 6 and hydroxy groups at positions 7 and 4'. It has been isolated from the mycelia of the fungus Cordyceps sinensis.
General Description
Glycitein is an isoflavone found in soy food products.
Biochem/physiol Actions
Glycitein is a soybean (yellow cultivar) isoflavonoid; its natural glycosides are synergistic with genistein in inducing specific gene expression. Glycitein may be used to study anti-oxidation processes at the level of gene transcription where it increases the binding of transcription factors [nuclear factor-E2-related factor 2 (Nrf2) and c-Jun] to the antioxidant response element (ARE) on HO-1 and NQO1 promoters. Glycitein may be used in combination with other isoflavonoids such as genistein and daidzein to study apoptosis.
Synthesis

124-63-0

79744-58-4
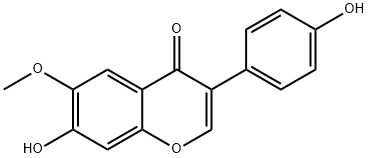
40957-83-3
Under nitrogen protection, boron trifluoride ethyl ether complex (1.6 mL, 13.13 mmol) was slowly added to an anhydrous N,N-dimethylformamide (10 mL) solution of deoxybenzene coupling (0.6 g, 2.19 mmol). After 15 min of reaction, anhydrous N,N-dimethylformamide (2 mL) solution of methanesulfonyl chloride (0.84 mL, 10.95 mmol) was added dropwise. The reaction mixture was heated and stirred in an oil bath at 70°C for 5 hours. Upon completion of the reaction, the mixture was cooled to room temperature and slowly poured into ice-cooled saturated aqueous sodium acetate solution (50 mL). The precipitated solid was collected by filtration and recrystallized from 70% ethanol to afford 7-hydroxy-3-(4-hydroxyphenyl)-6-methoxy-4H-benzopyran-4-one as a yellow solid (0.53 g, 85% yield) with a melting point of 336-338°C. The product structure was determined by 1H NMR. The structure of the product was confirmed by 1H NMR (300 MHz, DMSO-d6) and mass spectrometry (EI, 70 eV): 1H NMR δ 10.63 (br s, 1H, 7-OH), 9.54 (br s, 1H, 4'-OH), 8.27 (s, 1H, H-2), 7.41 (s, 1H, H-5), 7.37 (d, J = 8.7 Hz. 2H, H-2', 6'), 6.92 (s, 1H, H-8), 6.79 (d, J = 8.7 Hz, 2H, H-3', 5'), 3.86 (s, 3H, OCH3); mass spectra m/z 284 (M+, 24%), 283 (100), 268 (15), 255 (20), 212 (41), 171 (15).
References
[1] Patent: US2007/203227, 2007, A1. Location in patent: Page/Page column 3
Glycitein Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Chengdu GLP biotechnology Co Ltd | 028-87075086 13350802083 | scglp@glp-china.com | CHINA | 1824 | 58 |
| Hubei Danding Pharmaceutical Technology Co., Ltd | +86-18917019315 +86-13173601896 | 3582572959@qq.com | China | 496 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 20124 | 58 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 | zheyansh@163.com | CHINA | 3619 | 58 |
| Chengdu Greenpure Biopharma CO.,Ltd | 18283602253; +8618283602253 | jancyzheng@gcgreenpure.com | China | 954 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29820 | 58 |
| Chengdu Biopurify Phytochemicals Ltd. | +86-28-82633860; +8618080483897 | sales@biopurify.com | China | 3751 | 58 |
| Nanjing Dolon Biotechnology Co.,Ltd. | 18905173768 | 52513593@qq.com | CHINA | 2972 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19552 | 58 |
| Shanghai Standard Technology Co., Ltd. | 18502101150 | ft-sales@nature-standard.com | CHINA | 1923 | 58 |
View Lastest Price from Glycitein manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-22 | Glycitein
40957-83-3
|
US $50.00 / mL | 99.94% | 10g | TargetMol Chemicals Inc. | ||
 |
2025-09-25 | Glycitein
40957-83-3
|
US $8.00-1.00 / KG | 1KG | 99% | g-kg-tons, free sample is available | Henan Fengda Chemical Co., Ltd | |
 |
2023-02-24 | Glycitein
40957-83-3
|
US $0.00 / mg | 5mg | ≥98%(HPLC) | 10 g | Shanghai Standard Technology Co., Ltd. |