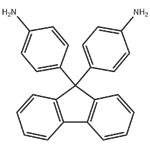
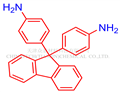
9,9-Bis(4-aminophenyl)fluorene
- CAS No.
- 15499-84-0
- Chemical Name:
- 9,9-Bis(4-aminophenyl)fluorene
- Synonyms
- FDA;BAFL;BAF/FDA;FDA/BAFL;AKOS BBB/416;Zinc03897005;Anilinefluorene;BIO-FARMA BF003152;TIMTEC-BB SBB008629;9,9-bis(4-aminopheny
- CBNumber:
- CB9439319
- Molecular Formula:
- C25H20N2
- Molecular Weight:
- 348.44
- MDL Number:
- MFCD00039156
- MOL File:
- 15499-84-0.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| 4,4′-(9-Fluorenylidene)dianiline 99% | 437913 | 25g | $276 |
| 9,9-Bis(4-aminophenyl)fluorene >98.0%(T) | B1549 | 25g | $110 |
| 9,9-Bis(4-aminophenyl)fluorene >98.0%(T) | B1549 | 100g | $330 |
| 9,9-Bis(4-aminophenyl)fluorene (purified by sublimation) >99.0%(HPLC)(T) | B2654 | 1g | $66 |
| 4,4''-(9H-Fluorene-9,9-diyl)dianiline | F591593 | 2.5g | $90 |
| More product size | |||
| Melting point | 237-239 °C (lit.) |
|---|---|
| Boiling point | 535.2±50.0 °C(Predicted) |
| Density | 1.245±0.06 g/cm3(Predicted) |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| Water Solubility | Insoluble in water |
| pka | 4.92±0.10(Predicted) |
| form | powder to crystal |
| color | White to Almost white |
| InChI | InChI=1S/C25H20N2/c26-19-13-9-17(10-14-19)25(18-11-15-20(27)16-12-18)23-7-3-1-5-21(23)22-6-2-4-8-24(22)25/h1-16H,26-27H2 |
| InChIKey | KIFDSGGWDIVQGN-UHFFFAOYSA-N |
| SMILES | C1(C2=CC=C(N)C=C2)(C2=CC=C(N)C=C2)C2=C(C=CC=C2)C2=C1C=CC=C2 |
| CAS DataBase Reference | 15499-84-0(CAS DataBase Reference) |
| UNSPSC Code | 12352100 |
| NACRES | NA.22 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302+H312+H332-H319-H335 | |||||||||
| Precautionary statements | P261-P280-P301+P312-P302+P352+P312-P304+P340+P312-P305+P351+P338 | |||||||||
| target organs | Respiratory system | |||||||||
| PPE | dust mask type N95 (US), Eyeshields, Gloves | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 20/21/22-36/37 | |||||||||
| Safety Statements | 26-36 | |||||||||
| WGK Germany | 3 | |||||||||
| TSCA | TSCA listed | |||||||||
| HazardClass | IRRITANT | |||||||||
| HS Code | 29215900 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| Hazard Classifications | Acute Tox. 4 Oral Eye Irrit. 2 STOT SE 3 |
|||||||||
| NFPA 704 |
|
9,9-Bis(4-aminophenyl)fluorene price More Price(25)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 437913 | 4,4′-(9-Fluorenylidene)dianiline 99% | 15499-84-0 | 25g | $276 | 2026-04-30 | Buy |
| TCI Chemical | B1549 | 9,9-Bis(4-aminophenyl)fluorene >98.0%(T) | 15499-84-0 | 25g | $110 | 2026-04-30 | Buy |
| TCI Chemical | B1549 | 9,9-Bis(4-aminophenyl)fluorene >98.0%(T) | 15499-84-0 | 100g | $330 | 2026-04-30 | Buy |
| TCI Chemical | B2654 | 9,9-Bis(4-aminophenyl)fluorene (purified by sublimation) >99.0%(HPLC)(T) | 15499-84-0 | 1g | $66 | 2026-04-30 | Buy |
| TRC | F591593 | 4,4''-(9H-Fluorene-9,9-diyl)dianiline | 15499-84-0 | 2.5g | $90 | 2021-12-16 | Buy |
9,9-Bis(4-aminophenyl)fluorene Chemical Properties,Uses,Production
Description
9,9-Bis(4-aminophenyl)fluorene (FDA) is a fluorene derivative with two aminophenyl substituents on the 9-position. This compound has partial conjugation and can be utilized as a dopant-free organic hole-transporting material in inverted perovskite solar cells. Due to its diaminophenyl groups, FDA is commonly employed in synthesizing polyimides that possess high thermal and hydrolytic stability for optical fiber light guide coatings. Because the FDA is not fully aromatized, it is chosen to produce thermally stable transparent polyimides. Additionally, the polyimide derived from 9,9-bis(4-aminophenyl)fluorene can be further modified through Friedel-Crafts alkylation on the fluorene moiety, as the polyimide backbone remains chemically stable.
Chemical Properties
White powder
Uses
9,9-bis(4-aminophenyl)fluorene be used for preparation of high-strength material and high-temperature polymers.
Preparation
The synthesis of 9,9-Bis(4-aminophenyl)fluorene can be carried out through the following stepwise procedure:
Prepare the reaction mixture:
Place 3.6 g of fluorenone (20 mmol), 7.2 g of aniline hydrochloride (55.6 mmol), 0.5 g of sodium bisulfite (4.8 mmol), 8.8 mL of aniline (96.4 mmol), and 6 mL of toluene in a 100 mL two-necked flask.
Ensure the reaction is conducted under nitrogen protection.
Add a water separator to the two-necked flask.
Begin the reaction:
Stir the reaction mixture and slowly increase the temperature to 120 °C.
Allow the reaction to continue for 30 minutes.
Separate the green liquid produced in the water separator.
Further reaction at elevated temperature:
Continue the reaction at 120 °C for an additional 3 hours.
Separate the water produced in the water separator.
Increase temperature and maintain reaction:
Slowly raise the temperature to 135 °C.
Keep the reaction at this temperature for 1.5 hours.
Monitor for completion:
Check the water separator for any further production of water.
Cease the reaction when no more water is produced in the water separator.
Cool down and adjust pH:
Allow the reacted material to cool to around 60 ℃.
Add 10 wt% potassium hydroxide solution to the reaction mixture.
Adjust the pH of the mixture to 9.
Stir the solution for 15 minutes while it is still hot.
Filtration and purification:
Filter the solution to remove any solid impurities.
Wash the filter cake with water.
Recrystallize the washed filter cake using 30 mL of toluene.
Filter the recrystallized product to obtain pure rice-white product.
Yield determination:
Weigh the obtained pure product, which should amount to 6.55 g.
Calculate the yield by dividing the obtained mass by the theoretical maximum mass and multiplying by 100%.
The yield of the pure rice-white product should be 94%.
Application
9,9-bis(4-aminophenyl)fluorene has good optical properties and semiconductor properties, and it is also an important intermediate for the synthesis of polyfluorene derivatives.
General Description
4,4′-(9-Fluorenylidene)dianiline is a diamine.
Synthesis

486-25-9
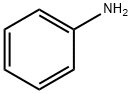
62-53-3

15499-84-0
General procedure for the synthesis of 9,9-bis(4-aminophenyl)fluorene from 9-fluorenone and aniline: 72 g of 9-fluorenone, 155.4 g of aniline hydrochloride, 10 g of sodium bisulfite and 72 g of 9-fluorenone were added to a reaction unit equipped with a water separator. Under stirring, 145 g of aniline and 100 g of toluene were heated to 125-130 °C and reacted at that temperature for 3 hours while the water generated was removed through the water separator. Subsequently, the temperature was slowly increased to 145-150 °C and maintained for 3 hours. Upon completion of the reaction, it was cooled to 95°C. At this temperature, a 10% sodium hydroxide solution was added dropwise until the pH of the reaction mixture was about 7. The neutralization reaction was carried out for 3 hours. Thermal stratification was carried out at 95°C to separate the aqueous layer and the upper oil layer was collected and transferred to a flask. The temperature of the mixture was gradually reduced to 20-30°C with stirring and stirred at this temperature for 1 hour to promote crystallization. After filtration, the mixture was dried to give about 185.6 g of crude product. 185.6 g of 9,9-bis(4-aminophenyl)fluorene and 300 g of xylene were added to the reactor and pulped at 30 °C and filtered to give 170 g of product in wet weight, which dried to 141 g. 141 g of 9,9-bis(4-aminophenyl)fluorene, 2800 g of deionized water, 49.3 g of 33% hydrochloric acid, and 28 g of activated charcoal were added to the flask and heated up to 65-70 °C , decolorized for 2 hours. The filtrate was hot-filtered at 70 °C and the filtrate was transferred to another flask, the pH was adjusted to 9-10 with 10% ammonia and stirred at 60-65 °C for 3 hours to ensure complete neutralization. After completion of neutralization, it was cooled to 20-30 °C, filtered and dried to give 130 g of product in 93.4% yield. The product was a pure white solid with a purity of 99.7%.
References
[1] PHAM H D, GIL-ESCRIG L, FERON K, et al. Boosting inverted perovskite solar cell performance by using 9,9-bis(4-diphenylaminophenyl)fluorene functionalized with triphenylamine as a dopant-free hole transporting material[J]. Journal of Materials Chemistry A, 2019, 20: 12507-12517. DOI:10.1039/C9TA01681C.
[2] G. SONG C. C. Colorless, heat resistant polyimide films derived from 2,3,3′,4′-biphenyltetracarboxylic dianhydride[J]. IOP Conference Series: Materials Science and Engineering, 2020, 73 1. DOI:10.1088/1757-899X/733/1/012035.
[3] WEI J, YU L, YAN L, et al. Synthesis of 9,9-bis(4-hydroxyphenyl) fluorene catalyzed by bifunctional ionic liquids[J]. RSC Advances, 2021, 52: 32559-32564. DOI:10.1039/D1RA05967J.
[4] SURASAK SEESUKPHRONRARAK. Synthesis and characterization of 9,9-bis(4-hydroxyphenyl and 4-aminophenyl)dibenzofluorenes: Novel fluorene-based monomers[J]. Journal of Polymer Science Part A: Polymer Chemistry, 2019, 57 24: 2602-2605. DOI:10.1002/pola.29540.
[5] HOU CHIEN CHANG. Synthesis of 9,9-bis(4-aminophenyl)fluorene-based benzoxazine and properties of its high-performance thermoset[J]. Journal of Polymer Science Part A: Polymer Chemistry, 2012, 50 11: 2201-2210. DOI:10.1002/pola.25993.
[6] Patent: CN107892649, 2018, A, . Location in patent: Paragraph 0023-0042
[7] Fortschr. Teerfarbenfabr. Verw. Industriezweige, vol. 23, p. 240
[8] DONGHUI YU. Fluorene-Based Phosphine Oxide Host Materials for Blue Electrophosphorescence: An Effective Strategy for a High Triplet Energy Level[J]. Chemistry - A European Journal, 2011, 17 9: 2592-2596. DOI:10.1002/chem.201003434.
9,9-Bis(4-aminophenyl)fluorene Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Shanghai Daeyeon Chemicals Co., Ltd | 021-64478606 +8615900664856 | daeyeon001@vip.163.com | China | 2062 | 58 |
| CHINATECH(TIANJIN) CHEMICAL CO.,LTD. | +86-86-18222270857 +8618222270857 | sales5@chinatechem.com | China | 75 | 58 |
| Jiangsu Ever Galaxy Chemical Co.,Ltd | +86-0527-81083685; +8618602515822 | info@egchemical.com | China | 79 | 58 |
| Henan Rane Chemical Co., Ltd. | +8618738066252 | info@ranechem.com | China | 2999 | 58 |
| Shanghai Hongzhixin Chemical Technology Co., Ltd | +86-021-58209276 +8613817242608 | selan@hzxchem.com | China | 59 | 58 |
| Chemport Science Technology Shanghai) Co., Ltd. | +undefined13816004845 | chen@chemport-sh.com | China | 188 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-86-13131129325 +8613131129325 | sales1@chuanghaibio.com | China | 5235 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 20124 | 58 |
| Hebei Zhuanglai Chemical Trading Co Ltd | +86-16264648883 | niki@zlchemi.com | China | 7245 | 58 |
| HANGZHOU HAILAN CHEMICAL CO.,LTD. | +86-57156122185 +86-15967590394 | sales@hailan-chem.com | China | 398 | 58 |
Related articles
- 9,9-Bis(4-aminophenyl)fluorene: Applications in Solar Cells, Polyimides, and Polymer Synthesis
- 9,9-Bis(4-aminophenyl)fluorene acts as a hole-transporter, polyimide precursor, and forms polymers via varied polymerizations ....
- Oct 17,2025
View Lastest Price from 9,9-Bis(4-aminophenyl)fluorene manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-30 | 9,9-Bis(4-aminophenyl)fluorene
15499-84-0
|
US $0.00 / kg | 1kg | 99%min | 20tons | HANGZHOU HAILAN CHEMICAL CO.,LTD. | |
 |
2026-01-20 | 9,9-Bis(4-aminophenyl) fluorene
15499-84-0
|
US $0.00 / kg | 25kg | ≥99% | ton | Zhongwei Coking Technology National Engineering Research Center Co., Ltd. | |
 |
2026-01-07 | 9,9-Bis(4-aminophenyl)fluorene (FDA)
15499-84-0
|
US $0.00-0.00 / kg | 1kg | ≥99.5% | 30TM | CHINATECH(TIANJIN) CHEMICAL CO.,LTD. |
-

- 9,9-Bis(4-aminophenyl)fluorene
15499-84-0
- US $0.00 / kg
- 99%min
- HANGZHOU HAILAN CHEMICAL CO.,LTD.
-

- 9,9-Bis(4-aminophenyl) fluorene
15499-84-0
- US $0.00 / kg
- ≥99%
- Zhongwei Coking Technology National Engineering Research Center Co., Ltd.
-

- 9,9-Bis(4-aminophenyl)fluorene (FDA)
15499-84-0
- US $0.00-0.00 / kg
- ≥99.5%
- CHINATECH(TIANJIN) CHEMICAL CO.,LTD.
15499-84-0(9,9-Bis(4-aminophenyl)fluorene)Related Search:
1of4





