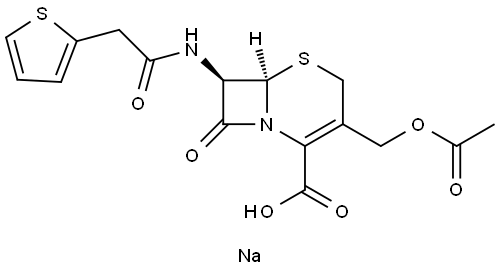
Cephalothin sodium
- CAS No.
- 58-71-9
- Chemical Name:
- Cephalothin sodium
- Synonyms
- 38253;Coaxin;Keflin;Seffin;Microtin;Averon 1;Cemastin;Lospoven;synclotin;Cephation
- CBNumber:
- CB9443527
- Molecular Formula:
- C16H16N2O6S2.Na
- Molecular Weight:
- 418.42
- MDL Number:
- MFCD00072025
- MOL File:
- 58-71-9.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Cefalotin for impurity B identification European Pharmacopoeia (EP) Reference Standard | Y0000505 | 10 mg | $127 |
| Cephalothin sodium salt 96.0-101.0% | C4520 | 1g | $154 |
| Cefalotin sodium European Pharmacopoeia (EP) Reference Standard | C0682000 | 170 mg | $167 |
| Cefalotin sodium British Pharmacopoeia (BP) Reference Standard | BP908 | 100 mg | $292 |
| Cephalothin sodium United States Pharmacopeia (USP) Reference Standard | 1102000 | 200mg | $488 |
| More product size | |||
| Melting point | 240°C |
|---|---|
| alpha | D +135° (c = 1.0 in water) |
| storage temp. | Inert atmosphere,2-8°C |
| solubility | H2O: 50 mg/mL, clear, faintly yellow |
| form | Solid |
| color | White to Off-White |
| Water Solubility | 158 mg/L |
| Merck | 13,1994 |
| BRN | 4120706 |
| Stability | Hygroscopic |
| InChI | InChI=1/C16H16N2O6S2.Na/c1-8(19)24-6-9-7-26-15-12(14(21)18(15)13(9)16(22)23)17-11(20)5-10-3-2-4-25-10;/h2-4,12,15H,5-7H2,1H3,(H,17,20)(H,22,23);/t12-,15-;/s3 |
| InChIKey | XXDPRVHYESWPEX-JQWYMVCGNA-N |
| SMILES | C(C1=C(CS[C@]2([H])[C@@H](C(N21)=O)NC(=O)CC1SC=CC=1)COC(=O)C)(=O)O.[Na] |&1:5,7,r| |
| CAS DataBase Reference | 58-71-9(CAS DataBase Reference) |
| FDA UNII | C22G6EYP8B |
| UNSPSC Code | 51282506 |
| NACRES | NA.85 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|---|---|
| Signal word | Danger |
| Hazard statements | H317-H334 |
| Precautionary statements | P261-P280-P284-P304+P340-P342+P311 |
| PPE | dust mask type N95 (US), Eyeshields, Faceshields, Gloves |
| Hazard Codes | Xn,Xi |
| Risk Statements | 42/43 |
| Safety Statements | 22-36/37 |
| WGK Germany | 2 |
| RTECS | XI0388300 |
| HS Code | 29419000 |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Resp. Sens. 1 Skin Sens. 1 |
| Toxicity | LD50 in mice, rats (mg/kg): >20000, >10000 orally; 5670, 7716 i.p. (Kuramoto) |
Cephalothin sodium price More Price(47)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | Y0000505 | Cefalotin for impurity B identification European Pharmacopoeia (EP) Reference Standard | 58-71-9 | 10 mg | $127 | 2026-04-30 | Buy |
| Sigma-Aldrich | C4520 | Cephalothin sodium salt 96.0-101.0% | 58-71-9 | 1g | $154 | 2026-04-30 | Buy |
| Sigma-Aldrich | C0682000 | Cefalotin sodium European Pharmacopoeia (EP) Reference Standard | 58-71-9 | 170 mg | $167 | 2026-04-30 | Buy |
| Sigma-Aldrich | BP908 | Cefalotin sodium British Pharmacopoeia (BP) Reference Standard | 58-71-9 | 100 mg | $292 | 2026-04-30 | Buy |
| Sigma-Aldrich | 1102000 | Cephalothin sodium United States Pharmacopeia (USP) Reference Standard | 58-71-9 | 200mg | $488 | 2026-04-30 | Buy |
Cephalothin sodium Chemical Properties,Uses,Production
Description
Cefalothin is a β-lactam cephalosporin antibiotic. It inhibits the growth of various Gram-positive and Gram-negative bacteria, including several strains of S. pyogenes, S. aureus, C. tetani, N. gonorrhoeae, Salmonella, and Shigella (MICs = 0.1-0.2, 0.312-0.625, 0.078, 1.25, 1.56-6.25, and 3.12-12.5 μg/ml, respectively). Cefalothin binds to E. coli penicillin-binding proteins (PBPs; IC50s = <0.25, 16, 37, and 1 μg/ml for PBP1a, 1bs, 2, and 3, respectively, in a radioligand binding assay), which interferes with bacterial morphogenesis. It exhibits antibacterial activity in mouse models of infection with S. pyogenes, D. pneumoniae, and S. aureus. Formulations containing cefalothin were previously used in the prophylaxis and treatment of bacterial infections.
Chemical Properties
Crystalline
Originator
Keflin,Lilly,US,1964
Uses
Cephalothin sodium salt is used to study the mechanism of liposome encapsulated antibiotics1, strategies for co-opting β-lactamases of Gram-negative bacteria for treatment of antibiotics2, and for immunology studies in relation to antibiotics.3 It is used to study the effect of expression, binding and inhibition of penicillin-binding proteins especially PBP6 on bacterial cell wall mucopeptide synthesis.
Uses
Antibacterial;Bacterial transpeptidase inhibitor
Uses
Cephalothin is a first-generation cephalosporin antibiotic used to study the mechanism of liposome encapsulated antibiotics,strategies for co-opting β-lactamases of Gram-negative bacteria for treatment of antibiotics,and for immunology studies in relation to antibiotics.It is used to study the effect of expression, binding and inhibition of penicillin-binding proteins especially PBP6 on bacterial cell wall mucopeptide synthesis.
Definition
ChEBI: Cephalothin sodium is an organic sodium salt. It contains a cefalotin.
Manufacturing Process
7-(2'-Thienylacetamido)cephalosporanic acid sodium salt may be produced from 2-thienylacetyl chloride, obtainable by treatment of 2-thienylacetic acid [Ernst, Berichte, 19 (1886) 3281] with thionyl chloride in a conventional manner. The 2-thienylacetyl chloride is then reacted with 7- aminocephalosporanic acid and then converted to the sodium salt using sodium hydroxide.
brand name
Keflin (Lilly); Seffin (GlaxoSmithKline).
Therapeutic Function
Antibacterial
General Description
Cephalothin, along with cephaloridine, was the first of the synthetic cephalosporin C class antibiotics to be introduced clinically. It was synthesized from 7-amino-cephalosporanic acid by Lilly Research Laboratories in 1962. Cephalothin shows strong activity against gram-positive and gram-negative bacteria and Leptospira, including benzylpenicillin-resistant strains. It has been used intravenously and intramuscularly to treat a variety of infections caused by Staphylococcus, Streptococcus, Escherichia coli, and Neisseria. The drug is metabolized in vivo, and the metabolite, deacetylcephalothin, is almost inactive.
Clinical Use
Cephalothin sodium (Keflin) occurs as a white to off-white,crystalline powder that is practically odorless. It is freelysoluble in water and insoluble in most organic solvents.Although it has been described as a broad-spectrum antibacterialcompound, it is not in the same class as the tetracyclines.Its spectrum of activity is broader than that ofpenicillin G and more similar to that of ampicillin. Unlikeampicillin, cephalothin is resistant to penicillinase producedby S. aureus and provides an alternative to the use ofpenicillinase-resistant penicillins for the treatment of infectionscaused by such strains.
Cephalothin is absorbed poorly from the GI tract andmust be administered parenterally for systemic infections. It is relatively nontoxic and acid stable. It is excreted rapidlythrough the kidneys; about 60% is lost within 6 hours of administration.Pain at the site of intramuscular injection andthrombophlebitis following intravenous injection have beenreported. Hypersensitivity reactions have been observed,and there is some evidence of cross-sensitivity in patientsnoted previously to be penicillin sensitive.
References
[1] W S BONIECE. In vitro and in vivo laboratory evaluation of cephalothin, a new broad spectrum antibiotic.[J]. Journal of Bacteriology, 1962, 84: 1292-1296. DOI: 10.1128/jb.84.6.1292-1296.1962
[2] N A CURTIS. Affinities of penicillins and cephalosporins for the penicillin-binding proteins of Escherichia coli K-12 and their antibacterial activity.[J]. Antimicrobial Agents and Chemotherapy, 1979, 16 5: 533-539. DOI: 10.1128/aac.16.5.533
Cephalothin sodium Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618092446649 | sarah@tnjone.com | China | 1143 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21590 | 55 |
| Xiamen AmoyChem Co., Ltd | +86-86-5926051114 +8615060885618 | sales@amoychem.com | China | 6369 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19552 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29794 | 58 |
| SIMAGCHEM CORP | +86-5922680277 +86-13806087780 | sale@simagchem.com | China | 17346 | 58 |
| Hebei Runbin Biotechnology Co. LTD | 13180553332 | 2179877681@qq.com | CHINA | 990 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354; +17819995354 | marketing@targetmol.com | United States | 32467 | 58 |
View Lastest Price from Cephalothin sodium manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-22 | Cephalothin sodium
58-71-9
|
US $0.00 / removed | 99.17% | 10g | TargetMol Chemicals Inc. | ||
 |
2026-04-22 | Cephalothin sodium
58-71-9
|
US $0.00 / removed | 99.17% | 10g | TargetMol Chemicals Inc. | ||
 |
2025-04-02 | Cephalothin sodium
58-71-9
|
US $0.00 / Kg | 1Kg | 99% | 10kg | Shaanxi TNJONE Pharmaceutical Co., Ltd |
-

- Cephalothin sodium
58-71-9
- US $0.00 / removed
- 99.17%
- TargetMol Chemicals Inc.
-

- Cephalothin sodium
58-71-9
- US $0.00 / removed
- 99.17%
- TargetMol Chemicals Inc.
-

- Cephalothin sodium
58-71-9
- US $0.00 / Kg
- 99%
- Shaanxi TNJONE Pharmaceutical Co., Ltd
58-71-9(Cephalothin sodium)Related Search:
1of4