- 24340-77-0
-
- $0.00 / 1g
-
2019-12-26
- CAS:24340-77-0
- Min. Order: 1g
- Purity: 95%min
- Supply Ability: 20kg/month
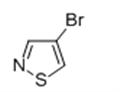
- 4-BROMO-ISOTHIAZOLE
-

- $1.00 / 1KG
-
2019-07-06
- CAS:24340-77-0
- Min. Order: 1KG
- Purity: 96%
- Supply Ability: 500kg
|
| | 4-BROMO-ISOTHIAZOLE Chemical Properties |
| Boiling point | 83.5±23.0 °C(Predicted) | | density | 1.857±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | form | solid | | pka | 1.59±0.22(Predicted) | | color | Light Yellow | | InChI | InChI=1S/C3H2BrNS/c4-3-1-5-6-2-3/h1-2H | | InChIKey | XAGKUQWKQVTDSK-UHFFFAOYSA-N | | SMILES | S1C=C(Br)C=N1 |
| | 4-BROMO-ISOTHIAZOLE Usage And Synthesis |
| Application | 4-Bromoisothiazol is a pharmaceutical intermediate that can be prepared by one-step bromination of isothiazole and can be used to prepare Bcr-Abl1 kinase inhibitors. | | Chemical Properties | White to off-white solid | | Synthesis | General procedure for the synthesis of 4-bromoisothiazole from isothiazole: Bromine (12.5 g, 78.12 mmol) was added slowly and dropwise to a solution of isothiazole (5.0 g, 58.82 mmol) in acetic acid (37 mL) over a period of 20 min at 95 °C. The reaction mixture was stirred continuously at 95 °C for 6 h and subsequently cooled to room temperature. The cooled mixture was poured into ice water (100 mL). The resulting mixture was extracted with ether (200 mL x 2). The organic phases were combined and washed with 6N sodium hydroxide solution to pH 7-8, followed by drying with anhydrous sodium sulfate and filtration. The filtrate was concentrated under reduced pressure and the resulting residue was purified by distillation to give 4-bromoisothiazole as a white solid (1.5 g, 15% yield). | | References | [1] Patent: WO2017/176961, 2017, A1. Location in patent: Paragraph 00375
[2] Journal of Medicinal Chemistry, 1968, vol. 11, # 1, p. 70 - 73 |
| | 4-BROMO-ISOTHIAZOLE Preparation Products And Raw materials |
|