|
|
| | (3,3-DIMETHYL-1-BUTYNE)DICOBALT HEXACARBONYL Basic information | | Uses |
| Product Name: | (3,3-DIMETHYL-1-BUTYNE)DICOBALT HEXACARBONYL | | Synonyms: | Dicobalt hexacarbonyl t-butylacetylene, 99%;(3,3-DIMETHYL-1-BUTYNE)DICOBALT HEXACARBONYL;(3,3-Dimethyl-1-butyne)dicobalthexacarbonyl,98%;Cobalt, hexacarbonyl[M-[(1,2-h:1,2-h)-3,3-diMethyl-1-butyne]]di-, (Co-Co) (9CI);(3,3-Dimethyl-1-butyne)dicobalt hexacarbonyl, 98% CCTBA;CCTBA;-1-butyne)dicobaL;t hexacarbonyL | | CAS: | 56792-69-9 | | MF: | C12H10Co2O6 | | MW: | 368.07 | | EINECS: | | | Product Categories: | metalocene | | Mol File: | 56792-69-9.mol | 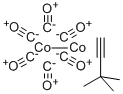 |
| | (3,3-DIMETHYL-1-BUTYNE)DICOBALT HEXACARBONYL Chemical Properties |
| Boiling point | 52°C / 0.8 | | form | liquid | | color | dark red | | Sensitive | air sensitive | | InChI | InChI=1S/C6H10.6CO.2Co/c1-5-6(2,3)4;6*1-2;;/h1H,2-4H3;;;;;;;; | | InChIKey | ZJCVAZZKJPVOPY-UHFFFAOYSA-N | | SMILES | C(C(C)(C)C)#C.[O+]#[C-].[O+]#[C-].[O+]#[C-].[O+]#[C-].[O+]#[C-].[O+]#[C-].[Co][Co] | | CAS DataBase Reference | 56792-69-9 |
| | (3,3-DIMETHYL-1-BUTYNE)DICOBALT HEXACARBONYL Usage And Synthesis |
| Uses |
- Substrate or catalyst for numerous organometallic transformations, including the Nicholas reaction, the Pauson– Khand reaction, alkyne cyclotrimerization, and alkyne hydrosilylation
- Precursor for the atomic layer deposition of Cobalt containing thin films
| | Application | (3,3-Dimethyl-1-butyne)dicobalt hexacarbonyl is an important precursor for the preparation of cobalt metal, cobalt nitride, cobalt silicides, etc., and exhibits excellent performance, especially for applications involving cobalt barrier layers/substrates, co-capping layers, and eutectic seed layers. In the case of barrier layers and seed layers, the cobalt film displays high conductivity in subsequent seed formation and electroplating steps. | | Synthesis | Under the protection of nitrogen atmosphere, cobalt carbonyl reagent and 1800 g of hydrocarbon solvent were added into the reactor, followed by temperature control at 10 °C. Then, 0.1 g of ionic liquid-modified graphene oxide material and 25 g of tert-butylacetylene were slowly added to the system. After the addition was completed, the mixture was stirred for reaction for 5 h under nitrogen atmosphere. After the reaction, filtration was carried out, and the obtained filtrate was distilled to remove volatile components, yielding the crude product of dicobalt hexacarbonyl (3,3-dimethyl-1-butyne), which was then purified to obtain pure (3,3-Dimethyl-1-butyne)dicobalt hexacarbonyl. |
| | (3,3-DIMETHYL-1-BUTYNE)DICOBALT HEXACARBONYL Preparation Products And Raw materials |
|