|
|
| | 5-FLUOROISOPHTHALIC ACID DIMETHYL ESTER Basic information |
| Product Name: | 5-FLUOROISOPHTHALIC ACID DIMETHYL ESTER | | Synonyms: | 5-FLUOROISOPHTHALIC ACID DIMETHYL ESTER;DIMETHYL 5-FLUOROISOPHTHALATE;dimethyl 5-fluorobenzene-1,3-dicarboxylate;1,3-Benzenedicarboxylic acid, 5-fluoro-, 1,3-dimethyl ester | | CAS: | 17449-48-8 | | MF: | C10H9FO4 | | MW: | 212.17 | | EINECS: | | | Product Categories: | | | Mol File: | 17449-48-8.mol | 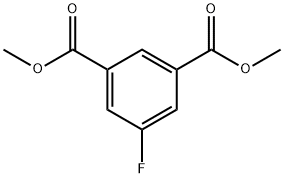 |
| | 5-FLUOROISOPHTHALIC ACID DIMETHYL ESTER Chemical Properties |
| Melting point | 56.5-57 °C | | Boiling point | 288.3±30.0 °C(Predicted) | | density | 1.252±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,Room Temperature | | Appearance | Off-white to light yellow Solid |
| | 5-FLUOROISOPHTHALIC ACID DIMETHYL ESTER Usage And Synthesis |
| Uses | Dimethyl 5-Fluoroisophthalate is used as a reactant in the preparation of quinazoline compounds as B-Raf inhibitors. | | Synthesis | General procedure for the synthesis of dimethyl 5-fluoroisophthalate from methanol and 5-fluoroisophthalic acid: to a solution of 5-fluoroisophthalic acid (1.3 g, 7.1 mmol) in methanol (30 ml) was added concentrated sulfuric acid (0.25 ml). The reaction mixture was heated to reflux for 12 hours. After completion of the reaction, methanol was removed by distillation under reduced pressure. The residue was neutralized with saturated sodium bicarbonate solution and subsequently extracted with dichloromethane. The organic phase was washed with saturated sodium chloride solution, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to give dimethyl 5-fluoroisophthalate 1.25 g (83% yield) as a white solid. Nuclear magnetic resonance hydrogen spectroscopy (NMR) data: δ 8.42 (s, 1H), 7.88 (d, 2H), 3.90 (s, 6H). | | References | [1] Patent: WO2007/71963, 2007, A2. Location in patent: Page/Page column 54 |
| | 5-FLUOROISOPHTHALIC ACID DIMETHYL ESTER Preparation Products And Raw materials |
|