- Borneol Oil
-

- $150.00 / 1kg
-
2025-05-23
- CAS:6627-72-1
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 500kg
- Borneol oil
-

- $0.00 / 1g
-
2025-02-13
- CAS:6627-72-1
- Min. Order: 1g
- Purity: 98%
- Supply Ability: 1000
|
| Product Name: | Borneol oil | | Synonyms: | DL-BORNEOL;1,7,7-TRIMETHYLNORBORNAN-2-OL;BORNEOL, (L);DL-2-Bornanol;(1S,4R,6R)-1,7,7-Trimethylbicyclo[2.2.1]heptan-6-ol;Natural borneol oil;6R)-1;7-Trimethylbicyclo[2.2.1]heptan-6-ol | | CAS: | 6627-72-1 | | MF: | C10H18O | | MW: | 154.25 | | EINECS: | 300-006-4 | | Product Categories: | API Intermediate | | Mol File: | 6627-72-1.mol | 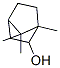 |
| | Borneol oil Chemical Properties |
| Melting point | 208°C | | Boiling point | 217.64°C (rough estimate) | | density | 1.0110 | | refractive index | 1.4586 (estimate) | | InChI | InChI=1S/C10H18O/c1-9(2)7-4-5-10(9,3)8(11)6-7/h7-8,11H,4-6H2,1-3H3 | | InChIKey | DTGKSKDOIYIVQL-UHFFFAOYSA-N | | SMILES | C1(O)C2(C)C(C)(C)C(CC2)C1 | | NIST Chemistry Reference | DL-Borneol(6627-72-1) |
| | Borneol oil Usage And Synthesis |
| Application | Borneol oil is typically used in small amounts in the scents of herbal camphor, as well as lavender, frankincense, cologne, and pine needle fragrances. In soap fragrances, it adds a refreshing aroma. It is also used in hygiene products, possessing anti-inflammatory and antibacterial properties, and is often combined with peppermint oil in tooth powder, prickly heat powder, clam oil, and pine needle-scented sprays for added fragrance. In edible applications, it can be used in trace amounts in nuts, chewing gum, and spicy flavorings. | | Chemical Properties | White or opaque hexagonal flaky crystals. Boiling point 208 ° C (left-handed), 214 ° C (right-handed), relative density 1.011-1.020, melting point 203-208 ° C, with ethanol and most of the essential oils are mutually soluble. The aroma is cool and sharp, slightly medicinal, woody and peppery. The aroma is ethereal, with a woody, slightly burnt and cool flavor. | | Definition | ChEBI: Borneol is a bornane monoterpenoid that is 1,7,7-trimethylbicyclo[2.2.1]heptane substituted by a hydroxy group at position 2. It has a role as a volatile oil component and a metabolite. | | Synthesis Reference(s) | The Journal of Organic Chemistry, 45, p. 1041, 1980 DOI: 10.1021/jo01294a024 | | Purification Methods | Crystallise borneol from pet ether (b 60-80o) and sublime it in vacuo.[Beilstein 6 II 81, 6 IV 281.] |
| | Borneol oil Preparation Products And Raw materials |
|