帕瑞昔布钠
| 中文名称 | 帕瑞昔布钠 |
|---|---|
| 中文同义词 | 帕瑞考昔钠;帕瑞昔布钠杂质(4个);帕瑞昔布钠;帕瑞西布纳原料药;帕瑞昔布(钠盐);N-[[4-(5-甲基-3-苯基-4-异恶唑基)苯基]磺酰]丙酰胺钠盐(帕来昔布钠);帕瑞昔布杂质25;帕瑞西布钠 |
| 英文名称 | PARECOXIB SODIUM |
| 英文同义词 | Parecoxib sodium (Intermediate in China ONLY);N-[4-(5-METHYL-3-PHENYLISOXAZOL-4-YL)PHENYLSULFONYL]PROPIONAMIDE, SODIUM SALT;Parecoxib sodiuM salt;Dynastat;N-[[4-(5-Methyl-3-phenyl-4-isoxazolyl)phenyl]sulfonyl]propanaMide SodiuM Salt;Rayzon;SC 69124A;PropanaMide,N-[[4-(5-Methyl-3-phenyl-4-isoxazolyl)phenyl]sulfonyl]-, sodiuM salt (1:1) |
| CAS号 | 198470-85-8 |
| 分子式 | C19H19N2NaO4S |
| 分子量 | 394.42 |
| EINECS号 | 813-689-5 |
| 相关类别 | 帕瑞考昔钠;原料药;医药原料药;医药原料;医药中间体;消炎镇痛;杂质对照品;医用原料;医药化工类;对照品-杂质对照品;Aromatics;Heterocycles;Intermediates & Fine Chemicals;Pharmaceuticals;科研试剂;药物杂质及中间体;化工原料药;原料;化工原料;073-帕瑞昔布钠;原料药及中间体;化工中间体;对照品 |
| Mol文件 | 198470-85-8.mol |
| 结构式 | 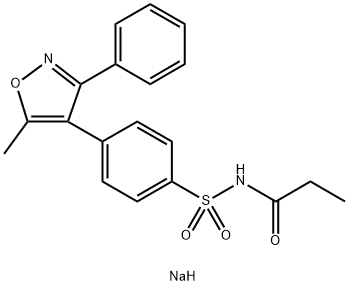 |
帕瑞昔布钠 性质
| 熔点 | 273-275°C |
|---|---|
| 储存条件 | Inert atmosphere,2-8°C |
| 溶解度 | DMF:10mg/mL;二甲基亚砜:15mg/mL;乙醇:3mg/mL; PBS(pH 7.2):5mg/mL |
| 形态 | 粉末 |
| 颜色 | 白色至米白色 |
| 水溶解性 | H2O: 2mg/mL, clear |
| InChI | InChI=1S/C19H18N2O4S.Na.H/c1-3-17(22)21-26(23,24)16-11-9-14(10-12-16)18-13(2)25-20-19(18)15-7-5-4-6-8-15;;/h4-12H,3H2,1-2H3,(H,21,22);; |
| InChIKey | OGSQVUBPCQXSGW-UHFFFAOYSA-N |
| SMILES | CC1ON=C(C2C=CC=CC=2)C=1C1C=CC(S(=O)(=O)NC(=O)CC)=CC=1.[NaH] |
帕瑞昔布钠药物对于疼痛,炎症和发热问题会有很大的帮助,可以选择性抑制降低组织包括内皮组织前列腺素的形成,不过对于血小板血栓烷素没有影响。帕瑞昔布钠在临床上一般用于中度或者重度手术后的急性疼痛的治疗。
帕瑞昔布钠具有抗炎,止痛的作用。帕瑞昔布是伐地昔布的前体药物,能选择性的抑制环氧化酶-2(COX-2),抑制花生四烯酸向前列腺素(PG)转化,抑制PG合成,从而发挥镇痛抗炎作用,而对COX-1抑制作用并不明显,因而在发挥镇痛、抗炎作用的同时,不影响胃肠道粘膜、血小板及肾功能。
|
COX-2
|
Parecoxib Sodium (0-200 μM; 24-48 hours) inhibits the cell proliferation of GBM cells in a dose-dependent manner in GBM cells.Parecoxib Sodium (200 μM; 24-48 hours) results in a decreased migratory ability of U343 cells than PBS-treated group.
Cell Viability Assay
| Cell Line: | GBM cells: U251 and U343 cells |
| Concentration: | 0 μM, 20 μM, 50 μM, 100 μM and 200 μM |
| Incubation Time: | 24-48 hours |
| Result: | Resulted in a slower BrdU incorporation rate of GBM cells including U251 and U343 cells. |
Parecoxib Sodium (intraperitoneal injection; 2.5, 5.0 or 10 mg/kg; once a day; 21 days) does not affect locomotor activity in the elevated plus-maze test, and Parecoxib at 5 and 10 mg/kg shows higher levels of percentage of time spent in the open arms.
| Animal Model: | Naive adult male ICR mice, 15 weeks old and weighing 25-35 g |
| Dosage: | 2.5 mg/kg, 5.0 mg/kg or 10 mg/kg |
| Administration: | Intraperitoneal injection; 2.5, 5.0 or 10 mg/kg; once a day; 21 days |
| Result: | Exerted an anxiolytic-like effect in the elevated plus-maze test. |
安全信息
| WGK Germany | WGK 1 |
|---|---|
| 存储类别 | 11 - 可燃固体 |
| 危险性类别 | 生殖毒性 类别2 |
| 更新日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
|---|---|---|---|---|---|
| 2026/03/03 | 46538 | 帕瑞昔布钠 Parecoxib sodium salt | 198470-85-8 | 1g | 3437元 |
| 2026/03/03 | S5418 | 帕瑞昔布钠 Parecoxib Sodium | 198470-85-8 | 25mg | 794.92元 |
