|
|
| | 6-Benzothiazolecarboxaldehyde(8CI,9CI) Basic information |
| Product Name: | 6-Benzothiazolecarboxaldehyde(8CI,9CI) | | Synonyms: | 6-Benzothiazolecarboxaldehyde(8CI,9CI);6-Benzothiazolecarboxaldehyde;6-Formylbenzothiazole;1,3-Benzothiazole-6-carboxaldehyde, 97%;6-Benzothiazolecarboxaldehyd;Benzo[d]thiazole-6-carbaldehyde;1,3-Benzothiazole-6-carbaldehyde;6-Benzothiazolecarbaldehyde | | CAS: | 19989-67-4 | | MF: | C8H5NOS | | MW: | 163.2 | | EINECS: | | | Product Categories: | BENZOTHIAZOLE | | Mol File: | 19989-67-4.mol | 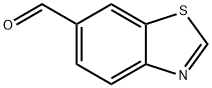 |
| | 6-Benzothiazolecarboxaldehyde(8CI,9CI) Chemical Properties |
| Boiling point | 311.4±15.0 °C(Predicted) | | density | 1.384 | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | form | Crystalline Powder or Low Melting Solid | | pka | -0.32±0.10(Predicted) | | color | White to yellow to brown |
| | 6-Benzothiazolecarboxaldehyde(8CI,9CI) Usage And Synthesis |
| Synthesis | Benzo[d]thiazol-6-ylmethanol (3.5 g, 21 mmol) was dissolved in anhydrous dichloromethane (20 mL) at room temperature, followed by addition of manganese dioxide (14 g, 0.21 mol). The reaction mixture was stirred continuously for 16 hours at room temperature. After completion of the reaction, the insoluble material was removed by filtration and the filtrate was processed by vacuum concentration to finally obtain benzothiazole-6-aldehyde (3.0 g, 88% yield) as a yellow solid. | | References | [1] Patent: WO2015/66371, 2015, A1. Location in patent: Paragraph 00368-00369
[2] Journal of the American Chemical Society, 2008, vol. 130, # 20, p. 6404 - 6410
[3] Journal of Medicinal Chemistry, 1968, vol. 11, # 2, p. 270 - 273 |
| | 6-Benzothiazolecarboxaldehyde(8CI,9CI) Preparation Products And Raw materials |
|