|
|
| | Benzenepropanoic acid,a-hydroxy-b-methoxy-b-phenyl-,(aS)- Basic information | | Uses |
| Product Name: | Benzenepropanoic acid,a-hydroxy-b-methoxy-b-phenyl-,(aS)- | | Synonyms: | Benzenepropanoic acid,a-hydroxy-b-methoxy-b-phenyl-,(aS)-;(S)-2-hydroxy-3-methoxy-3,3-diphenylpropanoic acid;(S)-2-Hydroxy-3-methoxy-3,3-diphenylpropionic acid;(S)-2-Hydroxyl -3-Methoxy-3,3-diphenylpropanoic acid;AMbrisentan InterMediate;Ambrisentan Hydroxy Acid Impurity;(S)-2-Hydroxy-3-methoxy-3,3-diphenylpropionic Acid;3-diphenylpropanoic acid | | CAS: | 178306-52-0 | | MF: | C16H16O4 | | MW: | 272.3 | | EINECS: | 1806241-263-5 | | Product Categories: | Drug Intermediates | | Mol File: | 178306-52-0.mol | 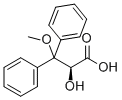 |
| | Benzenepropanoic acid,a-hydroxy-b-methoxy-b-phenyl-,(aS)- Chemical Properties |
| Melting point | 121 °C | | Boiling point | 430.3±45.0 °C(Predicted) | | density | 1.248 | | storage temp. | Sealed in dry,Room Temperature | | solubility | DMSO (Slightly), Ethanol (Sparingly, Sonicated), Methanol (Slightly) | | form | Solid | | pka | 3.24±0.28(Predicted) | | color | White to Off-White | | InChI | InChI=1/C16H16O4/c1-20-16(14(17)15(18)19,12-8-4-2-5-9-12)13-10-6-3-7-11-13/h2-11,14,17H,1H3,(H,18,19)/t14-/s3 | | InChIKey | RQJWOLFMWKZKCJ-WDHUHKLTNA-N | | SMILES | C(OC)(C1=CC=CC=C1)(C1C=CC=CC=1)[C@H](O)C(=O)O |&1:15,r| |
| | Benzenepropanoic acid,a-hydroxy-b-methoxy-b-phenyl-,(aS)- Usage And Synthesis |
| Uses | (S)-2-Hydroxy-3-methoxy-3,3-diphenylpropionic Acid is used in analytical studies for chiral discrimination of α-functionalized acids. Ambrisentan Hydroxy Acid Impurity. | | Chemical Properties | White powder | | Uses | (S)-2-Hydroxy-3-methoxy-3,3-diphenylpropionic Acid is used in analytical studies for chiral discrimination of α-functionalized acids. Ambrisentan Hydroxy Acid Impurity | | Synthesis | The general procedure for the synthesis of (S)-2-hydroxy-3-methoxy-3,3-diphenylpropionic acid from 2-hydroxy-3-methoxy-3,3-diphenylpropionic acid was as follows: 500 mg (1.84 mmol) of (RS)-2-hydroxy-3-methoxy-3,3-diphenylpropionic acid was dissolved in 8 mL of 2-propanol, followed by addition of 135 mg (0.92 mmol) of (S)-(+)-1,2,3,4-tetrahydro-1-naphthylamine. The reaction mixture was stirred under heated reflux conditions for 1 hour, then cooled to room temperature and continued stirring for 2 hours. The precipitated crystals were collected by filtration, washed with 2-propanol and finally dried under reduced pressure. The same operations as in Example 1 were performed according to the solvents listed in Table 1. | | References | [1] Patent: JP2017/128528, 2017, A. Location in patent: Paragraph 0005; 0041; 0042; 0045; 0046
[2] Patent: WO2012/17441, 2012, A1. Location in patent: Page/Page column 11-12
[3] Patent: CN104098462, 2017, B. Location in patent: Paragraph 0009; 0028; 0029; 0031
[4] Patent: JP5700378, 2015, B2. Location in patent: Paragraph 0071; 0072
[5] Patent: WO2013/57468, 2013, A1. Location in patent: Page/Page column 23-24 |
| | Benzenepropanoic acid,a-hydroxy-b-methoxy-b-phenyl-,(aS)- Preparation Products And Raw materials |
|