- Alendronic Acid
-

- $40.00 / 100mg
-
2026-03-06
- CAS:66376-36-1
- Min. Order:
- Purity: 99.89%
- Supply Ability: 10g
- Alendronic Acid
-
- $0.00 / 10mg
-
2025-12-16
- CAS:66376-36-1
- Min. Order: 10mg
- Purity: 98
- Supply Ability: 10000000
- Alendronic acid
-

- $0.00 / 25kg
-
2025-12-01
- CAS:66376-36-1
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10000KGS
|
| | Alendronic acid Basic information |
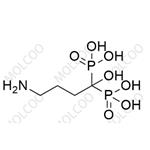
| Product Name: | Alendronic acid | | Synonyms: | 4-AMINO-1-HYDROXY-1,1-BUTYLIDINEDIPHOSPHONIC ACID;(4-AMINO-1-HYDROXY-1-PHOSPHONOBUTYL)PHOSPHONIC ACID;(4-AMINO-1-HYDROXYBUTYLIDENE)-BIS-(PHOSPHONIC ACID);(4-AMINO-1-HYDROXYBUTYLIDENE)DIPHOSPHONIC ACID;ALENDRONATE;ALENDRONIC ACID;(4-amino-1-hydroxybutylidene)bis-phosphonicaci;4-amino-1-hydroxybutylidene-1,1-bis(phosphonicacid) | | CAS: | 66376-36-1 | | MF: | C4H13NO7P2 | | MW: | 249.1 | | EINECS: | 204-352-8 | | Product Categories: | Miscellaneous Biochemicals;(intermediate of alendronate sodium) | | Mol File: | 66376-36-1.mol |  |
| | Alendronic acid Chemical Properties |
| Melting point | 230-235°C | | Boiling point | 616.7±65.0 °C(Predicted) | | density | 1.857±0.06 g/cm3(Predicted) | | storage temp. | Store at -20°C | | Water Solubility | Insoluble in water | | solubility | Aqueous Base (Sparingly), Methanol (Slightly), Water (Very Slightly) | | form | powder to crystal | | pka | pK1 2.72 ±0.05; pK2 8.73 ±0.05; pK3 10.5 ±0.1; pK4 11.6 ±0.1 at 25°, (0.1M KCl) | | color | White to Light yellow | | Merck | 14,229 | | Stability: | Hygroscopic | | EPA Substance Registry System | Phosphonic acid, P,P'-(4-amino-1-hydroxybutylidene)bis- (66376-36-1) |
| | Alendronic acid Usage And Synthesis |
| Chemical Properties | White to Light yellow powder to crystal. | | Originator | Adronat ,Neopharmed ,Italy | | Uses | Alendronate acid is an inhibitor of FDPS. | | Indications | Alendronic acid is a bisphosphonate used to treat and prevent osteoporosis and corticosteroid-induced osteoporosis. | | Preparation | Synthesis of Alendronic Acid: 1 mol of 4-aminobutyric acid was added to 1.5 mol of molten phosphoric acid at 95°C, followed by dropwise addition of 2 mol of phosphorus trihalide over 60 minutes. After the reaction mixture hardened, heating was continued for an additional 3 hours. The mixture was then hydrolyzed by adding 300 mL of water. Following cooling, the solution was poured into 1500 mL of methanol, and the resulting precipitate was collected to yield alendronic acid in 64.6% yield. | | Definition | ChEBI: Alendronic acid is a 1,1-bis(phosphonic acid) that is methanebis(phosphonic acid) in which the two methylene hydrogens are replaced by hydroxy and 3-aminopropyl groups. It has a role as an EC 2.5.1.1 (dimethylallyltranstransferase) inhibitor and a bone density conservation agent. It is a 1,1-bis(phosphonic acid) and a primary amino compound. It is a conjugate acid of an alendronate(1-). | | Manufacturing Process | 4-Amino-1-hydroxybutylidene-1,1-diphosphonic acid (ABDT).
Orthosphophorous acid (102.7 g; 1.25 moles) is introduced into a 2 liter-flask
with condenser, stirrer and dropping funnel, placed on a thermostatized bath;
the air is then removed with a nitrogen stream which is continued during all
the reaction. The acid is melted by heating the bath to 95°C. When melting is
complete, 4-aminobutyric acid (103.3; 1 mole) is added under stirring which
is continued till obtaining a doughy fluid. Phosphorous trihalide (176 ml; 2
moles) is added dropwise causing the mixture to boil and evolution of gaseous
hydrochloric acid which is damped by means of a suitable trap. The addition
rate is adjusted so as to keep a constant reflux for about 60 minutes. When
the addition is nearly over, the mixture swells, slowly hardening. Stirring is
continued as long as possible, whereafter the mixture is heated for further 3
hours. Without cooling, but removing the bath, water (300 ml) is added, first
slowly and then quickly. Heating and stirring are started again. Decolorizing
charcoal is added and the mixture is boiled for about 5 minutes, then hotfiltered
on paper and the filtrate is refluxed for 6 hours. After cooling is slowly
poured in stirred methanol (1500 ml) causing thereby the separation of a
white solid which collected and dried (161 g; 64.6%). The structure of ABDT
is confirmed by IR spectrum, proton magnetic and nuclear magnetic
resonance spectrum and elemental analysis.
The sodium salt of this acid may be prepared by adding of equivalent of
sodium hydroxide. | | Therapeutic Function | Antiosteoporotic | | Hazard | A reproductive hazard. | | Clinical Use | Bisphosphonate:
Treatment and prophylaxis of osteoporosis | | Drug interactions | Potentially hazardous interactions with other drugs
Calcium salts: reduced absorption of alendronate. | | Metabolism | Alendronate transiently distributes to soft tissues but is
then rapidly redistributed to bone or excreted in the urine.
There is no evidence that alendronate is metabolised in
animals or humans. Following a single intravenous dose of
[14C]-alendronate, approximately 50% of the radioactivity
was excreted in the urine within 72 hours and little or no
radioactivity was recovered in the faeces. | | References | [1] DÁVID ILLÉS NAGY. Synthesis of Hydroxymethylenebisphosphonic Acid Derivatives in Different Solvents.[J]. Molecules, 2016, 21 8. DOI:10.3390/molecules21081046.
[2] E. A. VOLOSNIKOVA. The synthesis of TNF-alpha conjugates with alendronic acid[J]. Russian Journal of Bioorganic Chemistry, 2017, 42 6: 638-645. DOI:10.1134/S1068162016060145.
[3] MOHAMED F. MADY* Malcolm A K Rocio Ortega. Exploring Modified Alendronic Acid as a New Inhibitor for Calcium-Based Oilfield Scales[J]. Energy & Fuels, 2022, 36 4: 1863-1873. DOI:10.1021/acs.energyfuels.1c03936. |
| | Alendronic acid Preparation Products And Raw materials |
|