DEMECOLCINE manufacturers
- Colcemid
-

- $31.00 / 1mg
-
2026-05-15
- CAS:477-30-5
- Min. Order:
- Purity: 99.65%
- Supply Ability: 10g
- DEMECOLCINE
-

- $1.00 / 1KG
-
2025-12-12
- CAS:477-30-5
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 420000KG
- DEMECOLCINE
-

- $8.00 / 1KG
-
2025-09-25
- CAS:477-30-5
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | DEMECOLCINE Basic information |
| Product Name: | DEMECOLCINE | | Synonyms: | substancef;Colcemide;DEMECOLCINE HYBRI-MAXR;DEMECOLCINE SOLUTION 10 UG/ML;Colcemid-D3=Demecolcine-D3;N-Deacetyl-N-methyl(deutero)colchicine;COLCEMID;COLCEMIDE(R) | | CAS: | 477-30-5 | | MF: | C21H25NO5 | | MW: | 371.43 | | EINECS: | 207-514-6 | | Product Categories: | All Inhibitors;Inhibitors | | Mol File: | 477-30-5.mol | 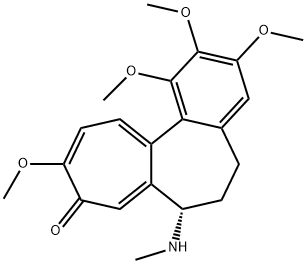 |
| | DEMECOLCINE Chemical Properties |
| Melting point | 73-75°C | | alpha | D20 -129.0° (c = 1 in chloroform) | | Boiling point | 501.11°C (rough estimate) | | density | 1.2350 (rough estimate) | | refractive index | 1.5614 (estimate) | | storage temp. | 2-8°C | | solubility | DMSO: 10 mg/mL | | form | powder | | pka | 8.48±0.40(Predicted) | | color | White to off-white | | Sensitive | Air & Light Sensitive | | BRN | 2822892 | | Stability: | Hygroscopic | | InChI | 1S/C21H25NO5/c1-22-15-8-6-12-10-18(25-3)20(26-4)21(27-5)19(12)13-7-9-17(24-2)16(23)11-14(13)15/h7,9-11,15,22H,6,8H2,1-5H3 | | InChIKey | NNJPGOLRFBJNIW-UHFFFAOYSA-N | | SMILES | N(C1CCc2c(c(c(c(c2)OC)OC)OC)C3=CC=C(C(=O)C=C31)OC)C |
| Hazard Codes | T,T+ | | Risk Statements | 25-26/27/28 | | Safety Statements | 22-24/25-45-36/37/39 | | RIDADR | UN 2811 6.1/PG 2 | | WGK Germany | 3 | | RTECS | GH0800000 | | F | 8-10-23 | | HazardClass | 6.1(a) | | PackingGroup | II | | Toxicity | LD50 oral in mouse: 25530ug/kg |
| | DEMECOLCINE Usage And Synthesis |
| Description | Colcemid is a colchicine derivative that inhibits tubulin polymerization as potently as colchicine (IC50 = 2.1 and 2.4 μM, respectively) but is less toxic. At very low (nanomolar) concentrations, colcemid suppresses microtubule dynamicity and inhibits cell migration, while at micromolar levels it blocks microtubule assembly, arresting cells in metaphase. Mitotic block by colcemid is used to synchronize cells and for karyotyping in cytogenetic studies. Prolonged exposure to colcemid can activate p53, leading to apoptosis. | | Chemical Properties | Faintly Yellow Crystalline Powder | | Uses | An antimitotic agent that disrupts microtubles by binding to tubulin and preventing its polymerization. Stimulates the intrinsic GTPase activity of tubulin. Induces apoptosis in several normal and tumor cell lines and activates the JNK/SAPK signaling pathway. | | Uses | Inhibitor of spindle fiber formation | | Uses | Cell synchronization agent; for chromosome visualization; to induce oocyte enucleation for somatic cell cloning. | | Definition | ChEBI: A secondary amino compound that is (S)-colchicine in which the N-acetyl group is replaced by an N-methyl group. Isolable from the autumn crocus, Colchicum autumnale, it is less toxic than
olchicine and is used as an antineoplastic. | | General Description | Colcemid is also known as demecolcine. Its generic name is N-methyl-N-deacetyl-colchicine. Colcemid depolymerizes microtubules and blocks mitosis at metaphase. | | Biochem/physiol Actions | Often in karyotyping and cell cycle research it is desirable to increase the yield of mitotic cells in a particular phase of the cell cycle. This can be achieved in a variety of ways with the most popular being the use of a cell cycle synchronizing agent such as demecolcine. Demecolcine will arrest cells in metaphase with no remarkable effect on the biochemical events in mitotic cells or in synchronized G1 and S phase cells. White blood cells are often treated with demecolcine to arrest cells in metaphase. | | Safety Profile | Poison by ingestion,
intraperitoneal, parenteral, intravenous, and
intramuscular routes. Human systemic
effects by ingestion: (skin and appendages)
hair effects. Human mutation data reported.
An experimental teratogen. Experimental
reproductive effects. When heated to
decomposition it emits toxic fumes of NOx. | | Purification Methods | Colcemide is purified by chromatography on silica and eluting with CHCl3/MeOH (9:1), and by recrystallisation from EtOAc/Et2O to form yellow prisms. UV in EtOH has max 243nm (� 30,200) and 350nm (� 16,3000). [Synthesis, IR, NMR, MS: Capraro & Brossi Helv Chim Acta 62 965 1979, Beilstein 8 IV 3319.] | | References | [1] ANJUM MUZAFFAR. Antitubulin effects of derivatives of 3-demethylthiocolchicine, methylthio ethers of natural colchicinoids, and thioketones derived from thiocolchicine. Comparison with colchicinoids[J]. Journal of Medicinal Chemistry, 1990, 33 2: 567-571. DOI: 10.1021/jm00164a015
[2] C L RIEDER R E P. Colcemid and the mitotic cycle.[J]. Journal of cell science, 1992, 102 ( Pt 3): 387-392. DOI: 10.1242/jcs.102.3.387
[3] MARY ANN JORDAN Leslie W. Microtubules as a target for anticancer drugs[J]. Nature Reviews Cancer, 2004, 4 4: 253-265. DOI: 10.1038/nrc1317
[4] JOZEF CURLEJ Peter C Jozef Bulla. Occurrence of chromosomal aneuploidy in rabbit oocytes and embryos at different developmental stages.[J]. Zygote, 2010, 18 3: 203-207. DOI: 10.1017/s0967199409990207
[5] HAILING YANG F C Anutosh Ganguly. Inhibition of cell migration and cell division correlates with distinct effects of microtubule inhibiting drugs.[J]. The Journal of Biological Chemistry, 2010: 32242-32250. DOI: 10.1074/jbc.m110.160820
[6] JANE BAYANI Jeremy A S. Preparation of Cytogenetic Specimens from Tissue Samples[J]. Current Protocols in Cell Biology, 2004, 23 1. DOI: 10.1002/0471143030.cb2202s23
[7] ANNA A SABLINA. p53 activation in response to microtubule disruption is mediated by integrin-Erk signaling[J]. Oncogene, 2001, 20 8: 899-909. DOI: 10.1038/sj.onc.1204156 |
| | DEMECOLCINE Preparation Products And Raw materials |
|