2,3-Diaminobenzotrifluoride 97% manufacturers
|
| | 2,3-Diaminobenzotrifluoride 97% Basic information |
| Product Name: | 2,3-Diaminobenzotrifluoride 97% | | Synonyms: | 3-(Trifluoromethyl)benzene-1,2-diamine;2,3-Diaminobenzotrifluoride 97%;3-(trifluoroMethyl)benzene-1;2,3-Diaminobenzotrifluoride, 3-(Trifluoromethyl)phenylene-1,2-diamine;3-(Trifluoromethyl)benzene-1,2-diamine 97%;3-(Trifluoromethyl)benzene-1,2-diamine97%;2,3-Diaminobenzotrifluoride97%;1,2-Benzenediamine, 3-(trifluoromethyl)- | | CAS: | 360-60-1 | | MF: | C7H7F3N2 | | MW: | 176.14 | | EINECS: | | | Product Categories: | | | Mol File: | 360-60-1.mol | 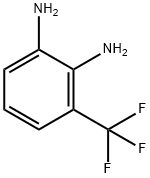 |
| | 2,3-Diaminobenzotrifluoride 97% Chemical Properties |
| Melting point | 41 °C | | Boiling point | 125 °C(Press: 14 Torr) | | density | 1.381±0.06 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2–8 °C | | pka | 2.94±0.10(Predicted) | | form | powder | | color | Yellow |
| | 2,3-Diaminobenzotrifluoride 97% Usage And Synthesis |
| Synthesis | Step 4: Synthesis of 3-trifluoromethyl-benzene-1,2-diamine; A suspension of 2-nitro-3-(trifluoromethyl)aniline (0.678 g, 3.2 mmol) formed with 10% palladium/carbon catalyst (0.067 g) in methanol (20 mL) was subjected to a hydrogenation reaction under a hydrogen atmosphere (using a hydrogen balloon) for 2 hours. Upon completion of the reaction, the catalyst was removed by filtration and the solvent was subsequently removed by evaporation under reduced pressure to afford 2,3-diaminobenzotrifluoride as an off-white solid (0.420 g, 73.17% yield). | | References | [1] Patent: WO2010/115736, 2010, A2. Location in patent: Page/Page column 102
[2] Journal of the Chemical Society, 1952, p. 4078
[3] Patent: US2005/54626, 2005, A1. Location in patent: Page/Page column 46
[4] Patent: US2010/120778, 2010, A1. Location in patent: Page/Page column 21
[5] Journal of Medicinal Chemistry, 2010, vol. 53, # 8, p. 3296 - 3304 |
| | 2,3-Diaminobenzotrifluoride 97% Preparation Products And Raw materials |
|