- 2-iodoaniline
-

- $0.00 / 1kg
-
2026-04-28
- CAS:615-43-0
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: Customise
- 2-Iodobenzenamine
-

- $29.00 / 1g
-
2026-04-22
- CAS:615-43-0
- Min. Order:
- Purity: 99.95%
- Supply Ability: 10g
- 2-Iodoaniline
-

- $10.00 / 1KG
-
2026-03-20
- CAS:615-43-0
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 5tons
|
| | 2-Iodoaniline Basic information |
| | 2-Iodoaniline Chemical Properties |
| Melting point | 55-58 °C (lit.) | | Boiling point | 262.0±23.0 °C(Predicted) | | density | 1.8155 (estimate) | | Fp | >230 °F | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | soluble in Chloroform, DCM, Ethyl Acetate, Toluene | | pka | 2.6(at 25℃) | | form | Crystalline Needles | | color | Yellow to brown | | Water Solubility | Insoluble in water. | | Sensitive | Light Sensitive | | BRN | 2204899 | | InChI | 1S/C6H6IN/c7-5-3-1-2-4-6(5)8/h1-4H,8H2 | | InChIKey | UBPDKIDWEADHPP-UHFFFAOYSA-N | | SMILES | Nc1ccccc1I | | CAS DataBase Reference | 615-43-0(CAS DataBase Reference) | | NIST Chemistry Reference | Benzenamine, 2-iodo-(615-43-0) | | EPA Substance Registry System | o-Iodoaniline (615-43-0) |
| Hazard Codes | Xn,Xi | | Risk Statements | 20/21/22-37/38-36/37/38 | | Safety Statements | 36/37-36/37/39-26-36 | | RIDADR | 2811 | | WGK Germany | 3 | | F | 8 | | Hazard Note | Irritant | | TSCA | TSCA listed | | HazardClass | 6.1(b) | | PackingGroup | III | | HS Code | 29214210 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | 2-Iodoaniline Usage And Synthesis |
| Chemical Properties | YELLOW TO BROWN CRYSTALLINE NEEDLES | | Uses | 2-Iodobenzenamine is an synthetic intermediate for the synthesis of complex organic compounds and pharmaceuticals. | | Uses | 2-Iodoaniline is used as an intermediate in organic synthesis. | | Preparation | 2-iodoaniline and its derivatives are commonly used for the preparation of indole-based anticancer drugs, which have high market value, and further synthesis can lead to high-end synthetic intermediates with complex structures.
Take a reaction tube, add 50mg of sodium azide, 75mg of 1-(2-iodophenyl)ethanol, 300uL of trifluoroacetic acid, 150uL of methanesulfonic acid and 1.0mL of hexane, and stir for 24 hours at 40℃. After the reaction, 10 mL of sodium hydroxide solution was added to quench the reaction. The organic phase was washed with 5 mL of brine, and the organic phase was combined and separated by column chromatography to obtain 58.4 mg of 2-iodoaniline in 89% yield.
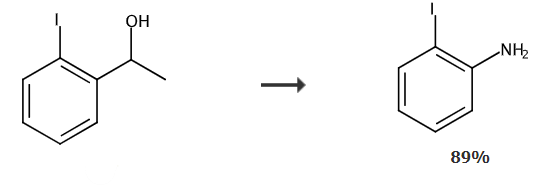
Synthesis of 2-iodoaniline | | Purification Methods | Distil 2-iodoaniline with steam and crystallise it from *benzene/pet ether. The N-acetyl derivative has m 110o. [Beilstein 12 IV 1542.] |
| | 2-Iodoaniline Preparation Products And Raw materials |
|