- L-165041
-

- $40.00 / 5mg
-
2026-04-20
- CAS:79558-09-1
- Min. Order:
- Purity: 97.97%
- Supply Ability: 10g
- L-165,041
-

- $1.00 / 1g
-
2019-12-27
- CAS:79558-09-1
- Min. Order: 1g
- Purity: 99%
- Supply Ability: 200kg
|
| | L-165,041 Basic information |
| Product Name: | L-165,041 | | Synonyms: | 4-[3-(2-PROPYL-3-HYDROXY-4-ACETYL)PHENOXY]PROPYLOXYPHENOXY-ACETIC ACID;4-[3-(4-ACETYL-3-HYDROXY-2-PROPYLPHENOXY)PROPOXY]PHENOXYACETIC ACID;L-165,041;COMPOUND P;L 1q5041;Acetic acid, 2-[4-[3-(4-acetyl-3-hydroxy-2-propylphenoxy)propoxy]phenoxy]-;2-(4-(3-(4-acetyl-3-hydroxy-2-propylphenoxy)propoxy)phenoxy)acetic acid;2-[4-[3-(4-acetyl-3-hydroxy-2-propylphenoxy)propoxy]phenoxy]- | | CAS: | 79558-09-1 | | MF: | C22H26O7 | | MW: | 402.44 | | EINECS: | | | Product Categories: | Intracellular receptor;API | | Mol File: | 79558-09-1.mol | 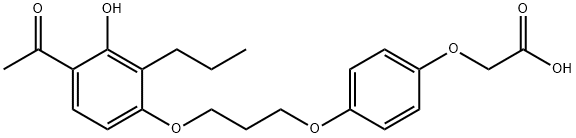 |
| | L-165,041 Chemical Properties |
| Melting point | 127-128 °C(lit.) | | Boiling point | 600.8±55.0 °C(Predicted) | | density | 1.222±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | solubility | DMSO: >10 mg/mL | | form | solid | | pka | 3.23±0.10(Predicted) | | color | off-white | | Stability: | Light Sensitive | | InChI | 1S/C22H26O7/c1-3-5-19-20(11-10-18(15(2)23)22(19)26)28-13-4-12-27-16-6-8-17(9-7-16)29-14-21(24)25/h6-11,26H,3-5,12-14H2,1-2H3,(H,24,25) | | InChIKey | HBBVCKCCQCQCTJ-UHFFFAOYSA-N | | SMILES | CCCc1c(O)c(ccc1OCCCOc2ccc(OCC(O)=O)cc2)C(C)=O |
| WGK Germany | 3 | | Storage Class | 11 - Combustible Solids |
| | L-165,041 Usage And Synthesis |
| Uses | L-165,041 has been used as a peroxisome proliferator-activated receptor β/δ (PPARβ/δ) ligand to study its influence on PPARβ/δ mediated postnatal myogenesis in C2C12 myoblasts. | | Definition | ChEBI: 2-[4-[3-(4-acetyl-3-hydroxy-2-propylphenoxy)propoxy]phenoxy]acetic acid is an aromatic ketone. | | General Description | A cell-permeable phenoxyacetic acid derivative that acts as a potent and selective peroxisome proliferator activator receptor δ (PPARδ) agonist (Ki = 6 nM for hPPARδ and 730 nM for hPPARγ). Potently induces adipocyte differentiation in NIH-PPARδ cells at 500 nM and raises total cholesterol in insulin resistant db/db mice without altering glucose or triglycerides levels. Increases UCP3 (uncoupling protein 3) gene expression in L6 myotubes. Inhibits cytokine-induced expression of VCAM-1 (vascular cell adhesion molecule-1) and the secretion of MCP-1 (monocyte chemotactic protein-1) in EAhy926 cells. | | Biological Activity | Potent PPAR δ agonist (K i = 6 nM); displays > 100-fold selectivity for both mouse and human PPAR δ receptors over other subtypes. In vivo, raises plasma cholesterol levels in insulin-resistant db/db mice and is neuroprotective in models of cerebral infarction and Parkinson's disease. | | Biochem/physiol Actions | Cell permeable: yes | | in vitro | l-165041, which is a selective and potent pparδligand, displayed in this specified transactivation system, apart from its highly efficacious pparδ agonist activity, partial and full agonism at, respectively, pparγ2 and pparαsubtypes [1]. | | in vivo | l-165041 could drastically reduce lipid accumulation in the mouse liver, decreasing total hepatic triglyceride and cholesterol content compared to the vehicle group. gene analysis demonstrated that l-165041 lowered hepatic expression of pparγ, apolipoprotein b, il-1β, and interleukin-6. in contrast, l-165041 increased hepatic expressions of pparδ, lipoprotein lipase, and atp-binding cassette transporter g1 (abcg1) [2]. | | target | PPARδ | | storage | Desiccate at RT | | references | [1] wurch t, junquero d, delhon a, pauwels j. pharmacological analysis of wild-type alpha, gamma and delta subtypes of the human peroxisome proliferator-activated receptor. naunyn schmiedebergs arch pharmacol. 2002 feb;365(2):133-40.
[2] lim hj, park jh, lee s, choi he, lee ks, park hy. ppardelta ligand l-165041 ameliorates western diet-induced hepatic lipid accumulation and inflammation in ldlr-/- mice. eur j pharmacol. 2009 nov 10;622(1-3):45-51. |
| | L-165,041 Preparation Products And Raw materials |
|