- Beta-Tocopherol
-

- $40.00 / 1mg
-
2026-04-29
- CAS:148-03-8
- Min. Order:
- Purity: 99.90%
- Supply Ability: 10g
|
| | β-Tocopherol Basic information |
| Product Name: | β-Tocopherol | | Synonyms: | 5,8-DIMETHYLTOCOL;RAC-BETA-TOCOPHEROL;beta-tocopherol;3,4-dihydro-2,5,8-trimethyl-2-(4,8,12-trimethyltridecyl)-2H-benzopyran-6-ol;Tocopherol,rac-b-;(2R)-2,5,8-trimethyl-2-(4,8,12-trimethyltridecyl)chroman-6-ol;rac-beta-Tocopherol (5,8-DiMethyltocol);β-Tocopherol (Racemic Mixture) | | CAS: | 148-03-8 | | MF: | C28H48O2 | | MW: | 416.68 | | EINECS: | 205-708-5 | | Product Categories: | TF - TOChromatography;Alphabetic;Food&Beverage Standards;Vitamins/CoFactors/Enzymes | | Mol File: | 148-03-8.mol | 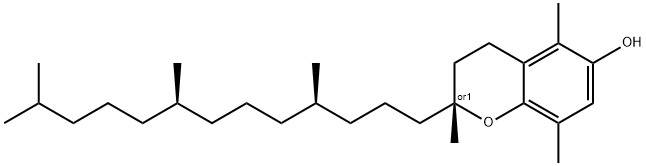 |
| | β-Tocopherol Chemical Properties |
| Melting point | <25℃ | | Boiling point | 474.94°C (rough estimate) | | density | 0.9882 (rough estimate) | | refractive index | 1.4480 (estimate) | | Fp | -23 °C | | storage temp. | 2-8°C | | solubility | Chloroform (Sparingly), Ethanol (Slightly), Ethyl Acetate (Slightly), Methanol ( | | form | Oil | | pka | 11.05±0.40(Predicted) | | color | Colourless to Yellow | | InChI | InChI=1/C28H48O2/c1-20(2)11-8-12-21(3)13-9-14-22(4)15-10-17-28(7)18-16-25-24(6)26(29)19-23(5)27(25)30-28/h19-22,29H,8-18H2,1-7H3/t21-,22-,28-/s3 | | InChIKey | WGVKWNUPNGFDFJ-UHFFFAOYSA-N | | SMILES | [C@@]1(C)(CCC[C@H](C)CCC[C@H](C)CCCC(C)C)OC2=C(C)C=C(O)C(C)=C2CC1 |&1:0,5,10,r| | | LogP | 10.724 (est) | | EPA Substance Registry System | 2H-1-Benzopyran-6-ol, 3,4-dihydro-2,5,8-trimethyl-2-[(4R,8R)-4,8,12-trimethyltridecyl]-, (2R)-rel- (148-03-8) |
| Hazard Codes | F,Xn,N | | Risk Statements | 11-38-48/20-51/53-62-67-65 | | Safety Statements | 36/37-61-62-16 | | RIDADR | UN1208 3/PG 2 | | WGK Germany | 3 | | TSCA | TSCA listed | | Storage Class | 3 - Flammable liquids | | Hazard Classifications | Acute Tox. 3 Dermal
Acute Tox. 3 Inhalation
Acute Tox. 3 Oral
Flam. Liq. 2
STOT SE 1 |
| | β-Tocopherol Usage And Synthesis |
| Description | (±)-β-Tocopherol is a lipid-soluble form of vitamin E with antioxidant activity. It scavenges 2,2-diphenyl-1-picrylhydrazyl (DPPH; ) radicals with an EC50 value of 68.2 μM. (±)-β-Tocopherol (100 μM) inhibits LPS-induced COX-2 gene expression in mouse RAW 264.7 macrophage cells. Unlike (±)-α-tocopherol , (±)-β-tocopherol does not inhibit smooth muscle cell proliferation, decrease PKC activity, increase phosphoprotein phosphatase 2A (PP2A) activity, or modify expression of the α-tropomyosin gene. [Matreya, LLC. Catalog No. 1071] | | Uses | One of the naturally occurring forms of Vitamin E. Is biologically less active than α-Tocopherol. This is the racemic mixture of all stereoisomers. | | Definition | ChEBI: Beta-tocopherol is a tocopherol in which the chroman-6-ol core is substituted by methyl groups at positions 5 and 8. While it is found in low concentrations in many vegetable oils, only cottonseed oil contains significant amounts. It has a role as a plant metabolite and a food component. It is a vitamin E and a tocopherol. | | References | [1] REGINA BRIGELIUS-FLOHé Maret G T. Vitamin E: function and metabolism[J]. FASEB Journal, 1999, 13 10: 1145-1155. DOI: 10.1096/fasebj.13.10.1145
[2] CHIY-RONG CHEN. Tocopherols and Triterpenoids from Sida acuta[C]//54 1. 2013: 41-45. DOI: 10.1002/jccs.200700008
[3] YUKIO MURAKAMI. Inhibitory effects of tocopherols on expression of the cyclooxygenase-2 gene in RAW264.7 cells stimulated by lipopolysaccharide, tumor necrosis factor-α or Porphyromonas gingivalis fimbriae.[J]. In vivo, 2013, 27 4: 451-458.
|
| | β-Tocopherol Preparation Products And Raw materials |
|