1,4,7-tris-Boc-1,4,7,10-tetraaza-cyclododecane manufacturers
- Tris-BOC-Cyclen
-
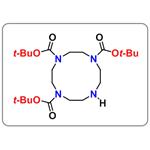
- $0.00 / 5g
-
2025-06-07
- CAS:175854-39-4
- Min. Order: 5g
- Purity: >95.00%
- Supply Ability: 5g
|
| | 1,4,7-tris-Boc-1,4,7,10-tetraaza-cyclododecane Basic information |
| Product Name: | 1,4,7-tris-Boc-1,4,7,10-tetraaza-cyclododecane | | Synonyms: | 1,4,7-tris-Boc-1,4,7,10-tetraaza-cyclododecane;1,4,7-Tri-Boc-1,4,7,10-tetraazacyclododecane;N,N',N''-tris-Boc-cyclen;1,4,7,10-Tetraazacyclododecane-1,4,7-tricarboxylic acid, tris(1,1-diMethylethyl) ester;TriBoc-Cyclen;Tri-tert-butyl 1,4,7,10-tetraazacyclotetradecane-1,4,7-tricarboxylate;Tri-tert-Butyl 1,4,7,10-tetraazacyclododecane-1,4,7-tricarboxylate;1,4,7,10-Tetraazacyclododecane-1,4,7-tricarboxylic acid, 1,4,7-tris(1,1-dimethylethyl) ester | | CAS: | 175854-39-4 | | MF: | C23H44N4O6 | | MW: | 472.62 | | EINECS: | | | Product Categories: | | | Mol File: | 175854-39-4.mol | 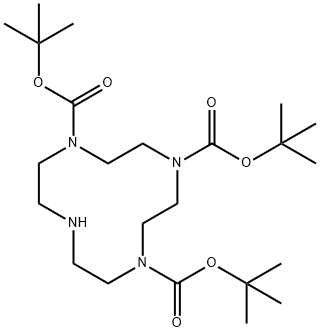 |
| | 1,4,7-tris-Boc-1,4,7,10-tetraaza-cyclododecane Chemical Properties |
| Melting point | 72-73℃ | | Boiling point | 542.8±35.0 °C(Predicted) | | density | 1.065 | | storage temp. | 2-8°C | | form | powder | | pka | 9.52±0.20(Predicted) | | Sensitive | Hygroscopic | | InChI | InChI=1S/C23H44N4O6/c1-21(2,3)31-18(28)25-12-10-24-11-13-26(19(29)32-22(4,5)6)15-17-27(16-14-25)20(30)33-23(7,8)9/h24H,10-17H2,1-9H3 | | InChIKey | ZXASHTYJQJMCQR-UHFFFAOYSA-N | | SMILES | N1(C(OC(C)(C)C)=O)CCNCCN(C(OC(C)(C)C)=O)CCN(C(OC(C)(C)C)=O)CC1 |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26 | | WGK Germany | 3 | | HS Code | 29339900 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | 1,4,7-tris-Boc-1,4,7,10-tetraaza-cyclododecane Usage And Synthesis |
| Chemical Properties | White powder | | Uses | Building block used to attach the Cyclen azacrown to a molecular scaffold, building block for a trifold cyclen Zn complex as phosphate dianion receptor in aqueous solution | | Synthesis | A solution of chloroform (10 mL) of di-tert-butyl dicarbonate (1.77 g, 8.12 mmol) was slowly added dropwise through a constant pressure dropping funnel to a solution of chloroform (40 mL) containing rotundin (0.51 g, 2.90 mmol) and triethylamine (1.25 mL, 8.99 mmol). The reaction mixture was stirred at room temperature overnight and then concentrated by rotary evaporator. The resulting crude product was purified by silica gel column chromatography using gradient elution from ethyl acetate to 5% methanol/dichloromethane to afford the target compound 1,4,7-tri-tert-butoxycarbonyl-1,4,7,10-tetraazacyclododecane as a white solid (1.02 g, 80% yield). The product characterization data were in agreement with literature reports. | | References | [1] Journal of Materials Chemistry, 2012, vol. 22, # 36, p. 18784 - 18787
[2] Journal of the American Chemical Society, 2004, vol. 126, # 30, p. 9248 - 9256
[3] RSC Advances, 2016, vol. 6, # 28, p. 23645 - 23652
[4] Journal of Organic Chemistry, 2005, vol. 70, # 1, p. 115 - 123
[5] Chemical Communications, 2011, vol. 47, # 21, p. 6036 - 6038 |
| | 1,4,7-tris-Boc-1,4,7,10-tetraaza-cyclododecane Preparation Products And Raw materials |
|