|
|
| Product Name: | SHA 68 | | Synonyms: | SHA 68;N-(4-fluorobenzyl)-3-oxo-1,1-diphenyltetrahydro-1H-oxazolo[3,4-a]pyrazine-7(3H)-carboxamide;N-[(4-Fluorophenyl)methyl]tetrahydro-3-oxo-1,1-diphenyl-3H-oxazolo[3,4-a]pyrazine-7(1H)-carboxamide;3H-Oxazolo[3,4-a]pyrazine-7(1H)-carboxamide, N-[(4-fluorophenyl)methyl]tetrahydro-3-oxo-1,1-diphenyl-;N-(4-Fluorobenzyl)-3-oxo-1,1-diphenyltetrahydro-3H-oxazolo[3,4-a]pyrazine-7(1H)-carboxamide;N-[(4-fluorophenyl)methyl]-3-oxo-1,1-diphenyl-hexahydro-1H-[1,3]oxazolo[3,4-a]pyrazine-7-carboxamide | | CAS: | 847553-89-3 | | MF: | C26H24FN3O3 | | MW: | 445.49 | | EINECS: | | | Product Categories: | | | Mol File: | 847553-89-3.mol | 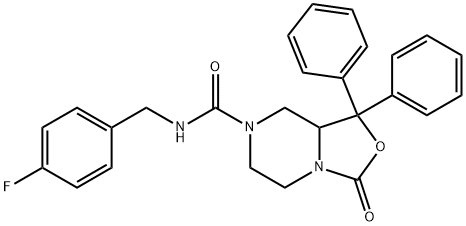 |
| | SHA 68 Chemical Properties |
| Boiling point | 727.8±60.0 °C(Predicted) | | density | 1.35±0.1 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | DMF: 16 mg/ml; DMSO: 14 mg/ml; Ethanol: 20 mg/ml; Ethanol:PBS (pH 7.2)(1:2): 0.33 mg/ml | | form | A crystalline solid | | pka | 13.32±0.40(Predicted) | | color | White to off-white | | InChI | InChI=1S/C26H24FN3O3/c27-22-13-11-19(12-14-22)17-28-24(31)29-15-16-30-23(18-29)26(33-25(30)32,20-7-3-1-4-8-20)21-9-5-2-6-10-21/h1-14,23H,15-18H2,(H,28,31) | | InChIKey | SFRQIPRTNYHJHP-UHFFFAOYSA-N | | SMILES | C12C(C3=CC=CC=C3)(C3=CC=CC=C3)OC(=O)N1CCN(C(NCC1=CC=C(F)C=C1)=O)C2 |
| WGK Germany | WGK 3 | | Storage Class | 11 - Combustible Solids |
| | SHA 68 Usage And Synthesis |
| Description | SHA-68 is an antagonist of the neuropeptide S receptor (NPSR; IC50s = 22 and 23.8 nM for the NPSR Asn107 and NPSR Ile107 isoforms, respectively). It is selective for NPSR over a panel of 14 G protein-coupled receptors exhibiting no activity at a concentration of 10 μM. SHA-68 (50 mg/kg) reduces NPS-induced horizontal activity and vertical rearing and climbing in mice. SHA-68 also reduces conditioned reinstatement of cocaine seeking in rats. | | Uses | SHA 68 is a selective neuropeptide S antagonist. | | in vivo | SHA 68 (i.p.; 5 and 50 mg/kg) reduces NPS-induced horizontal activity and vertical rearing and climbing[1].
SHA 68 (i.v.; 1 mg/kg) has a T1/2 of 0.74 hours, a CL of 4.29 mL/min/kg, and a Vss of 2.53 L/kg[1].
SHA 68 (i.p.; 2.5 mg/kg) has a T1/2 of 0.43 hours, a CL of 4.56 mL/min/kg[1].
| Animal Model: | Male C57BL/6 mice age 8-12 weeks[1] | | Dosage: | 5 and 50 mg/kg | | Administration: | i.p. | | Result: | Reduced NPS-induced horizontal activity and vertical rearing and climbing.
|
| Animal Model: | Male C57BL/6 mice age 8-12 weeks[1] | | Dosage: | 1 mg/kg (Pharmacokinetic Analysis) | | Administration: | i.p. | | Result: | Had a T1/2 of 0.74 hours, a CL of 4.29 mL/min/kg, and a Vss of 2.53 L/kg. |
| | storage | Store at +4°C | | References | [1] NAOE OKAMURA. Synthesis and pharmacological in vitro and in vivo profile of 3-oxo-1,1-diphenyl-tetrahydro-oxazolo[3,4-a]pyrazine-7-carboxylic acid 4-fluoro-benzylamide (SHA 68), a selective antagonist of the neuropeptide S receptor.[J]. Journal of Pharmacology and Experimental Therapeutics, 2008, 325 3: 893-901. DOI: 10.1124/jpet.107.135103
[2] MARSIDA KALLUPI. Neuropeptide S facilitates cue-induced relapse to cocaine seeking through activation of the hypothalamic hypocretin system.[J]. Proceedings of the National Academy of Sciences of the United States of America, 2010: 19567-19572. DOI: 10.1073/pnas.1004100107 |
| | SHA 68 Preparation Products And Raw materials |
|