|
|
| | N-(3-Aminophenyl)methanesulfamide Basic information |
| Product Name: | N-(3-Aminophenyl)methanesulfamide | | Synonyms: | N-(3-AMINO PHENYL)METHANE SULFONAMIDE;m-Aminomethylsulfonylaniline;n-(3-aminophenyl)methanesulfon;N-(3-Aminophenyl)methanesulfamide;3-(Methylsulfonamido)aniline;N-methylsulfonyl-m-phenylenediamine;N-(3-Aminophenyl)methanesulfonamide ,97%;N-(3-Aminophenyl) | | CAS: | 37045-73-1 | | MF: | C7H10N2O2S | | MW: | 186.23 | | EINECS: | | | Product Categories: | | | Mol File: | 37045-73-1.mol | 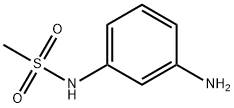 |
| | N-(3-Aminophenyl)methanesulfamide Chemical Properties |
| Melting point | 117-121 °C (lit.) | | Boiling point | 361.8±44.0 °C(Predicted) | | density | 1.408±0.06 g/cm3(Predicted) | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | pka | 9.12±0.10(Predicted) | | InChI | InChI=1S/C7H10N2O2S/c1-12(10,11)9-7-4-2-3-6(8)5-7/h2-5,9H,8H2,1H3 | | InChIKey | UQRRCQRFQGOHAI-UHFFFAOYSA-N | | SMILES | CS(NC1=CC=CC(N)=C1)(=O)=O |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36 | | WGK Germany | 3 | | HazardClass | IRRITANT | | HS Code | 29350090 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | N-(3-Aminophenyl)methanesulfamide Usage And Synthesis |
| Chemical Properties | Light red solid | | Uses | Reactant for:
- Preparation of biologically and pharmacologically active molecules
| | Synthesis | The general procedure for the synthesis of m-aminomethanesulfonanilide from N-(methylsulfonyl)-3-nitroanilide was as follows: N-(3-nitrophenyl)methanesulfonamide (2.6 g, 12.0 mmol, 1.00 equiv), iron powder (3.38 g, 60.5 mmol, 5.03 equiv) and ammonium chloride (6.48 g, 121 mmol, 10.1 equiv) were dissolved in a solvent mixture of methanol (40 mL) and water (20 mL). The reaction mixture was heated at 70 °C for the reaction. After completion of the reaction, the insoluble material was removed by filtration through diatomaceous earth. The filtrate was concentrated under reduced pressure, diluted with ethyl acetate, washed twice sequentially with saturated saline and the organic phase was dried with anhydrous sodium sulfate. Finally, it was purified by column chromatography (silica gel 115 g, eluent: 20% to 40% ethyl acetate/hexane) to give 1.55 g of orange solid product in 69% yield. | | References | [1] Patent: WO2014/6066, 2014, A1. Location in patent: Paragraph 0235
[2] Patent: WO2012/59932, 2012, A1. Location in patent: Page/Page column 149-150
[3] ChemMedChem, 2014, vol. 9, # 5, p. 962 - 972
[4] Patent: US2663732, 1950, |
| | N-(3-Aminophenyl)methanesulfamide Preparation Products And Raw materials |
|