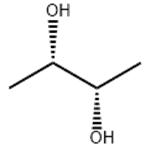
- (S,S)-2,3-Butanediol
-
- $0.00 / 10g
-
2026-01-04
- CAS:19132-06-0
- Min. Order: 0.1g
- Purity: >97.0%(GC)
- Supply Ability: 500kg
- (S S) -2 3-Butanediol
-

- $5.00 / 1KG
-
2025-05-26
- CAS:19132-06-0
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10000kg
- L-(+)-2,3-Butanediol
-

- $15.00 / 1KG
-
2021-07-02
- CAS:19132-06-0
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
|
| Product Name: | (S,S)-2,3-Butanediol | | Synonyms: | (S-(R,R))-butane-2,3-diol;(2S,3S)-(+)-2,3-BUTANEDIOL;(2S,3S)-2,3-BUTANEDIOL;2,3-BUTANEDIOL, [S-(R*,R*)]-;L(+)-2,3-BUTANEDIOL;(S,S)-(+)-2,3-DIHYDROXYBUTANE;(S,S)-(+)-2,3-BUTANEDIOL;(S,S)-2,3-BUTANEDIOL | | CAS: | 19132-06-0 | | MF: | C4H10O2 | | MW: | 90.12 | | EINECS: | | | Product Categories: | organic alcohol;Chiral Compounds;Diols;Analytical Chemistry;Chiral Building Blocks;e.e. / Absolute Configuration Determination (NMR);Enantiomer Excess & Absolute Configuration Determination;Simple Alcohols (Chiral);Synthetic Organic Chemistry;Chiral Compound;Chiral Building Blocks;Organic Building Blocks;Polyols;19132-06-0 | | Mol File: | 19132-06-0.mol |  |
| | (S,S)-2,3-Butanediol Chemical Properties |
| Melting point | 20°C | | alpha | 13 º (c=neat) | | Boiling point | 179-182 °C (lit.) | | density | 0.987 g/mL at 25 °C (lit.) | | refractive index | n20/D 1.433 | | Fp | 185 °F | | storage temp. | 2-8°C | | solubility | Chloroform (Slightly), Methanol (Slightly) | | pka | 14.67±0.20(Predicted) | | form | Oily Liquid | | color | Slightly yellow | | Specific Gravity | 0.987 | | PH | 9-10 (500g/l, H2O, 20℃) | | Optical Rotation | [α]20/D +13°, neat | | explosive limit | 3.1-11.4%(V) | | Water Solubility | soluble | | BRN | 1718899 | | Stability: | hygroscopic | | InChI | 1S/C4H10O2/c1-3(5)4(2)6/h3-6H,1-2H3/t3-,4-/m0/s1 | | InChIKey | OWBTYPJTUOEWEK-IMJSIDKUSA-N | | SMILES | C[C@H](O)[C@H](C)O | | LogP | -0.655 (est) | | CAS DataBase Reference | 19132-06-0(CAS DataBase Reference) | | NIST Chemistry Reference | 2,3-Butanediol, [S-(R*,R*)]-(19132-06-0) |
| Hazard Codes | Xi | | Safety Statements | 24/25 | | RIDADR | NA 1993 / PGIII | | WGK Germany | 3 | | HS Code | 29053995 | | Storage Class | 10 - Combustible liquids |
| | (S,S)-2,3-Butanediol Usage And Synthesis |
| Description | 2, 3-Butanediol, which is very important for a variety of chemical feedstocks and liquid fuels, can be derived from the bioconversion of natural resources1-2. It may be used in the production of antifreeze solvents, extraction solvents, butadiene, polymers, polyurethane and diacetyl and may also be used as a fuel1, 2. It can be used as a precursor in the manufacture of a range of chemical products, various plastics and pesticides, including the solvents methyl ethyl ketone (MEK), gamma-butyrolactone (GBL), and 1, 3-butadiene3. Methyl ethyl ketone can be used as a liquid fuel additive. Many other derivatives for potential uses as antifreeze agent (levo-form of 2,3 BDL), solvents, and plastic can also be prepared from 2.3-BDL. Furthermore, it can be added as a flavoring agent in food products when converted to a diacetyl by dehydrogenation2. Nowadays, it has been more and more applied of microbial fermentation for the production of 2.3-BDL2,4.
| | Reference |
- https://www.sigmaaldrich.com/catalog/product/sial/18967?lang=en®ion=US
- Lee, Jinwon, et al. "Biological production of 2,3-butanediol." Applied Microbiology & Biotechnology 55.1(2001):10-8.
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3147483/
- Oliver, John W. K., et al. "Cyanobacterial conversion of carbon dioxide to 2, 3-butanediol. PNAS 110.4(2013):1249-1254.
| | Chemical Properties | SLIGHTLY YELLOW OILY LIQUID | | Uses | (S,S)-2,3-Butanediol is the (S,S)-isomer of 2,3-Butanediol (B690020), a naturally occurring compound found in cocoa butter. | | Definition | ChEBI: The (S,S) diastereoisomer of butane-2,3-diol. | | General Description | L-(+)-2,3-Butanediol is a stereoisomer of 2,3-Butanediol (2,3-BD). 2,3-BD may be used in the production of antifreeze solvents, extraction solvents, butadiene, polymers, polyurethane and diacetyl and may also be used as a fuel. | | Synthesis | (1) 6.06 g of the ketoreductase-containing solution obtained in step 1 above was placed in a clean 250 mL single-necked flask equipped with a magnet with 20 mL of phosphate buffer salt solution at a concentration of 0.01 mol/L, and the single-necked flask was placed in a water bath at 20 C. The solution in the single-necked flask was stirred and mixed at a speed of 400 rpm, and a mixture was obtained. (2) During the stirring process, the temperature of the water bath was kept constant, and 0.202 g of nicotinamide adenine dinucleotide, 100 mL of isopropanol, and 20.2 g of 2,3-butanedione were sequentially added to the above mixture for an airtight reaction, and a reaction solution was obtained. (3) The above reaction solution was extracted and filtered under reduced pressure to obtain a light yellow filtrate, and then the filtrate was subjected to rotary evaporation treatment at 45 C. and 0.08 MPa, and the extract was obtained by waiting for almost no liquid to flow out. (4) The above extract was placed in a water bath at 50C, and a distillation column and condenser tube were used to raise the temperature without liquid to 70C for distillation and separation, and (2S,3S)-(+)-2,3-butanediol could be obtained. | | Purification Methods | Purify it by fractional distillation. The bis-(4-nitrobenzoate) has m 141-142o and [�] (-) or (+) 52o (c 4 CHCl3). [Ghirardelli & Lucas J Am Chem Soc 79 734 1957, Rubin et al. J Am Chem Soc 74 425 1952, Neish Can J Res 27 6 1949, Neish & Ledingham Can J Res 27 694 1949, Beilstein 1 IV 2524-2525.] |
| | (S,S)-2,3-Butanediol Preparation Products And Raw materials |
|