| Company Name: |
Chengdu RefMedic Biotech Co Ltd Gold
|
| Tel: |
028-82253651 18140204969 |
| Email: |
sales@refmedic.com |
| Products Intro: |
Product Name:γ-sanshool
CAS:78886-65-4
Purity:>98% (HPLC)10mg/支 Package:5mg;10mg;20mg;50mg;100mg;1g
|
| Company Name: |
Chengdu Push Bio-Technology Co., Ltd.
|
| Tel: |
028-85370565-229 18080489829 |
| Email: |
18080489829@163.com |
| Products Intro: |
Product Name:γ-sanshool
CAS:78886-65-4
Purity:95% Package:10mg
|
| Company Name: |
Chengdu Peter-like Biotechnology Co., Ltd.
|
| Tel: |
18108052721 18108052721; |
| Email: |
2850505130@qq.com |
| Products Intro: |
Product Name:γ-Sanshooel
CAS:78886-65-4
Purity:HPLC>=98% Package:5mg;10mg;20mg;50mg;100mg;200mg
|
| Company Name: |
Shanghai Hewu Biotechnology Co., LTD
|
| Tel: |
021-57886085 17317861055 |
| Email: |
shhebio@126.com |
| Products Intro: |
Product Name:γ-sanshool
CAS:78886-65-4
Purity:HPLC>98% Package:10mg 20mg 100mg 500mg 1g 10g Remarks:HPLC
|
|
| | γ-sanshool Basic information |
| Product Name: | γ-sanshool | | Synonyms: | γ-Sanshooel;γ-sanshool;2,4,8,10,12-Tetradecapentaenamide, N-(2-methylpropyl)-, (2E,4E,8Z,10E,12E)-;γ-Sanshool | | CAS: | 78886-65-4 | | MF: | C18H27NO | | MW: | 273.41 | | EINECS: | | | Product Categories: | | | Mol File: | 78886-65-4.mol | 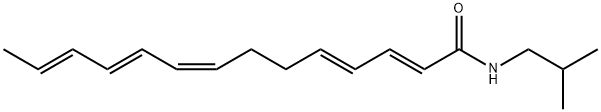 |
| | γ-sanshool Chemical Properties |
| Boiling point | 460.2±38.0 °C(Predicted) | | density | 0.911±0.06 g/cm3(Predicted) | | form | Solid | | pka | 14.67±0.46(Predicted) | | color | White to off-white | | InChI | InChI=1S/C18H27NO/c1-4-5-6-7-8-9-10-11-12-13-14-15-18(20)19-16-17(2)3/h4-9,12-15,17H,10-11,16H2,1-3H3,(H,19,20)/b5-4+,7-6+,9-8-,13-12+,15-14+ | | InChIKey | KVUKDCFEXVWYBN-JDXPBYPHSA-N | | SMILES | C(NCC(C)C)(=O)/C=C/C=C/CC/C=C\C=C\C=C\C |
| | γ-sanshool Usage And Synthesis |
| Uses | γ-Sanshool can be isolated from Zanthoxylum piperitum. γ-Sanshool inhibits human ACAT-1 and ACAT-2 activities with IC50s of 12.0 and 82.6 μM[1]. | | Definition | ChEBI: Gamma-sanshool is an enamide obtained by the formal condensation of 2-methylpropanamine with tetradeca-2,4,8,10,12-pentaenoic acid (the 2E,4E,8Z,10E,12E stereoisomer). Isolated from Zanthoxylum piperitum, it exhibits inhibitory activity against acyl-CoA:cholesterol acyltransferase. It has a role as an EC 2.3.1.26 (sterol O-acyltransferase) inhibitor and a plant metabolite. It is an enamide and a secondary carboxamide. It is functionally related to a 2-methylpropanamine. | | References | [1] Park YD, et al. Human acyl-CoA: cholesterol acyltransferase inhibitory activities of aliphatic acid amides from Zanthoxylum piperitum DC. Biol Pharm Bull. 2007 Jan;30(1):205-7. |
| | γ-sanshool Preparation Products And Raw materials |
|