20587-30-8
 20587-30-8 结构式
20587-30-8 结构式
基本信息
5-甲基-2-硝基苯甲酸甲酯(易制爆,甄准不供应)
5-Methyl-2-Nitrobenzoate Methyl
Methyl 5-Methyl-2-Nitrobenzoate
6-Nitro-m-toluic Acid Methyl Ester
Methyl 5-Methyl-2-nitrobenzoate >
5-Methyl-2-nitrobenzoic acid methyl ester
Benzoic acid, 5-methyl-2-nitro-, methyl ester
5-methyl-2-nitrobenzoic acid methyl ester 90%
2-(Ethoxycarbonyl)-4-methylnitrobenzene, 3-(Ethoxycarbonyl)-4-nitrotoluene
2-(Methoxycarbonyl)-4-methylnitrobenzene, 3-(Methoxycarbonyl)-4-nitrotoluene
物理化学性质
制备方法
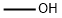
67-56-1

3113-72-2

20587-30-8
将5-甲基-2-硝基苯甲酸(8g,44.198mmol)溶于甲醇(80mL)中,并将溶液冷却至0℃。在搅拌下,缓慢滴加硫酸(5mL)。随后,将反应混合物加热至90℃,并在该温度下持续搅拌16小时。反应进程通过薄层色谱(TLC,展开剂为50%乙酸乙酯/己烷)进行监测。反应完成后,减压蒸馏除去过量的甲醇。将残余物倒入水(150mL)中,并用乙酸乙酯(150mL×2)进行萃取。合并有机相,依次用饱和碳酸氢钠溶液(50mL×2)和盐水(20mL)洗涤。有机层经无水硫酸钠干燥后,过滤并浓缩,得到5-甲基-2-硝基苯甲酸甲酯(产率:8g,93%)。产物结构经1H NMR(400MHz,CDCl3)确认:δ7.86-7.84(d,1H,J = 8Hz),7.48(s,1H),7.40-7.38(d,1H,J = 8Hz),3.91(s,3H),3.47(s,3H)。
参考文献:
[1] Journal of Heterocyclic Chemistry, 1984, vol. 21, p. 1807 - 1816
[2] Patent: WO2015/38417, 2015, A1. Location in patent: Paragraph 151
[3] Bulletin of the Korean Chemical Society, 2011, vol. 32, # 12, p. 4309 - 4315
[4] Bioorganic and Medicinal Chemistry, 2006, vol. 14, # 7, p. 2209 - 2224
[5] Chemische Berichte, 1909, vol. 42, p. 433
