N-BOC-1,6-diaminohexane
- CAS No.
- 51857-17-1
- Chemical Name:
- N-BOC-1,6-diaminohexane
- Synonyms
- tert-Butyl (6-aMinohexyl)carbaMate;N-BOC-1,6-DIAMINOHEXANE;N-BOC-1,6-HEXANEDIAMINE;TERT-BUTYL N-(6-AMINOHEXYL)CARBAMATE;tert-butyl (6-aMinohexyl)carbaMate hydrochloride;AURORA KA-7567;AURORA KA-7545;AURORA KA-4392;Boc-NH-(CH2)6-NH2;TIMTEC-BB SBB006530
- CBNumber:
- CB0702254
- Molecular Formula:
- C11H24N2O2
- Molecular Weight:
- 216.32
- MDL Number:
- MFCD00671489
- MOL File:
- 51857-17-1.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| N-Boc-1,6-hexanediamine ≥98.0% (NT) | 79229 | 1g | $56.1 |
| N-Boc-1,6-hexanediamine ≥98.0% (NT) | 79229 | 5g | $197 |
| N-(tert-Butoxycarbonyl)-1,6-diaminohexane >97.0%(GC)(T) | A1375 | 1g | $26 |
| N-(tert-Butoxycarbonyl)-1,6-diaminohexane >97.0%(GC)(T) | A1375 | 5g | $85 |
| N-(tert-Butoxycarbonyl)-1,6-diaminohexane >97.0%(GC)(T) | A1375 | 25g | $361 |
| More product size | |||
| Boiling point | 106-110 °C (0.3 mmHg) |
|---|---|
| Density | 0.965 g/mL at 20 °C(lit.) |
| refractive index |
n |
| Flash point | 125 °C |
| storage temp. | Keep in dark place,Inert atmosphere,2-8°C |
| solubility | Soluble in dichloromethane and ethyl acetate. |
| pka | 12.93±0.46(Predicted) |
| form | Viscous Liquid |
| color | Clear light yellow |
| Sensitive | Air Sensitive |
| BRN | 2089264 |
| InChI | InChI=1S/C11H24N2O2/c1-11(2,3)15-10(14)13-9-7-5-4-6-8-12/h4-9,12H2,1-3H3,(H,13,14) |
| InChIKey | RVZPDKXEHIRFPM-UHFFFAOYSA-N |
| SMILES | C(OC(C)(C)C)(=O)NCCCCCCN |
| CAS DataBase Reference | 51857-17-1(CAS DataBase Reference) |
| UNSPSC Code | 12352116 |
| NACRES | NA.22 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS05 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H314 | |||||||||
| Precautionary statements | P280-P303+P361+P353-P304+P340+P310-P305+P351+P338-P363-P405 | |||||||||
| PPE | Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter | |||||||||
| Hazard Codes | C | |||||||||
| Risk Statements | 34 | |||||||||
| Safety Statements | 26-36/37/39-45 | |||||||||
| RIDADR | UN 2735 8/PG 2 | |||||||||
| WGK Germany | 3 | |||||||||
| F | 10-34 | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29241900 | |||||||||
| Storage Class | 8A - Combustible corrosive hazardous materials | |||||||||
| Hazard Classifications | Eye Dam. 1 Skin Corr. 1B |
|||||||||
| NFPA 704 |
|
N-BOC-1,6-diaminohexane price More Price(37)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 79229 | N-Boc-1,6-hexanediamine ≥98.0% (NT) | 51857-17-1 | 1g | $56.1 | 2026-03-19 | Buy |
| Sigma-Aldrich | 79229 | N-Boc-1,6-hexanediamine ≥98.0% (NT) | 51857-17-1 | 5g | $197 | 2026-03-19 | Buy |
| TCI Chemical | A1375 | N-(tert-Butoxycarbonyl)-1,6-diaminohexane >97.0%(GC)(T) | 51857-17-1 | 1g | $26 | 2026-03-19 | Buy |
| TCI Chemical | A1375 | N-(tert-Butoxycarbonyl)-1,6-diaminohexane >97.0%(GC)(T) | 51857-17-1 | 5g | $85 | 2026-03-19 | Buy |
| TCI Chemical | A1375 | N-(tert-Butoxycarbonyl)-1,6-diaminohexane >97.0%(GC)(T) | 51857-17-1 | 25g | $361 | 2026-03-19 | Buy |
N-BOC-1,6-diaminohexane Chemical Properties,Uses,Production
Chemical Properties
Off-White Solid
Uses
N-Boc-1,6-diaminohexane is used to prepare 1,3-Bis[6-(Boc-amino)hexyl]urea by reacting with carbonyl dichloride in the presence of triethylamine. Further, it is used as a reagent for the introduction of a C6-spacer.
Application
N-Boc-1,6-hexanediamine can be used as a linear hexyl spacer (C6-spacer) to synthesize:
Biodegradable poly(disulfide amine)s for gene delivery.
A multifunctional dendrimer for theranostics.
Polyamide platinum anti-cancer complexes designed to target cancer specific DNA sequences.
Self-assembled monolayers (SAMs) that resist adsorption of proteins.
[N-(6-(4-Hydroxy-6-isopropylamino-1,3,5-triazin-2-ylamino)hexyl)-5-hydroxy-1,4-naphthoquinone-3-propionamide] (JUG-HATZ), which can be used in designing electrochemical immunosensors.
Preparation
synthesis of N-BOC-1,6-diaminohexane: 1,6-Diaminohexane 3 (Fig. 1) (100 g, 0.86 mol) was dissolved in CH2Cl2 (300 ml) and cooled in an ice bath to 0–3°C. To the stirred solution 0.3 eq. di-tert-butyl bicarbonate (62.6 g, 0.29 mol) was added slowly over a period of 1 h. The reaction was allowed to warm up to r.t., and proceeded overnight. The reaction mixture was extracted with saturated aqueous NaHCO3 (50 ml, three times). The organic phases were pooled, dried, and evaporated under reduced pressure. The resulting oil was dissolved in 200 ml, 1 N HCl and extracted with ether. The aqueous phase was washed with ether, made basic to a pH of 10 with aqueous 2 N NaO3, and extracted with ethyl acetate. The organic phases were pooled, dried, and evaporated under reduced pressure. The resulting oil was dissolved in 200 ml, 1 N HCL and extracted with ether. The aqueous phase was washed with ether, made basic to a pH of 10 with aqueous 2 N NaOH, and extracted with ethyl acetate. The organic extracts were pooled, dried over Na2SO4 and evaporated to give 10.5 g of homogeneous N-BOC-1, 6- diaminohexane 2c (Fig. 1) as a yellow oil which was used without further purification.
TLC Rf 0.51; 1H NMR (CDCL3) d 4.54 (bs, 1H; NH), 3.05 1 2.62 (m, 4H; NCH2), 1.41 (m, 9H; CH3), 1.29 (m, 8H; CH2).
Evaluation of aminoalkylmethacrylate nanoparticles as colloidal drug carrier systems. Part I: synthesis of monomers, dependence of the physical properties on the polymerization methods
reaction suitability
reagent type: cross-linking reagent
Synthesis

124-09-4
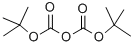
24424-99-5

51857-17-1
GENERAL STEPS: 1,6-Diaminohexane (20.0 g, 172 mmol) was suspended in 50 mL of dichloromethane (DCM) at 0 °C. To this suspension was slowly added a solution of di-tert-butyl dicarbonate (Boc2O, 3.75 g, 17.2 mmol) dissolved in 24 mL of DCM. After addition, the ice bath was removed and the reaction mixture was allowed to warm up to room temperature with continuous stirring for 18 hours. After completion of the reaction, the precipitate was removed by filtration and the filtrate was washed sequentially with saturated aqueous sodium bicarbonate (3 x 50 mL), water (2 x 50 mL) and brine (50 mL). The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated to give an oily product tert-butyl N-(6-aminohexyl)carbamate 3.1 g in 84% yield. The product was characterized by 1HNMR (300 MHz, CDCl3): δ 4.56 (s, 1H), 3.09 (dd, J=12.6,6.3Hz, 2H), 2.83-2.58 (t, 2H), 1.66-1.17 (m, 17H).
References
[1] Tetrahedron Letters, 2008, vol. 49, # 16, p. 2527 - 2532
[2] Journal of Materials Chemistry B, 2014, vol. 2, # 5, p. 502 - 510
[3] Bioorganic and Medicinal Chemistry Letters, 2010, vol. 20, # 23, p. 6956 - 6959
[4] Journal of Labelled Compounds and Radiopharmaceuticals, 2009, vol. 52, # 5, p. 139 - 145
[5] Bioorganic and Medicinal Chemistry, 2006, vol. 14, # 19, p. 6570 - 6580
N-BOC-1,6-diaminohexane Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21592 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 33024 | 60 |
| Jinan Carbotang Biotech Co.,Ltd. | +8615866703830 | figo.gao@foxmail.com | China | 8497 | 58 |
| Accela ChemBio Inc. | +1-858-6993322 | info@accelachem.com | United States | 21193 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29794 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354; +17819995354 | marketing@targetmol.com | United States | 32467 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34563 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 | 1026@dideu.com | China | 9731 | 58 |
View Lastest Price from N-BOC-1,6-diaminohexane manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
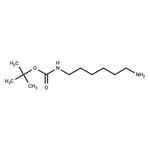 |
2026-04-17 | NH2-C6-NH-Boc
51857-17-1
|
US $29.00 / g | 99.97% | 10g | TargetMol Chemicals Inc. | ||
 |
2021-08-12 | N-tert-Butoxycarbonyl-1,6-hexanediamine
51857-17-1
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd | |
 |
2019-12-20 | N-tert-Butoxycarbonyl-1,6-hexanediamine
51857-17-1
|
US $1.00 / ASSAYS | 1ASSAYS | 97-99% | 1000KGS | Career Henan Chemical Co |
-

- NH2-C6-NH-Boc
51857-17-1
- US $29.00 / g
- 99.97%
- TargetMol Chemicals Inc.
-

- N-tert-Butoxycarbonyl-1,6-hexanediamine
51857-17-1
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd
-

- N-tert-Butoxycarbonyl-1,6-hexanediamine
51857-17-1
- US $1.00 / ASSAYS
- 97-99%
- Career Henan Chemical Co
51857-17-1(N-BOC-1,6-diaminohexane)Related Search:
1of4






