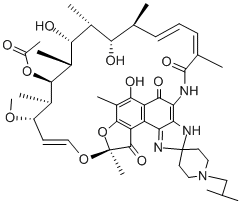
Rifabutin
- CAS No.
- 72559-06-9
- Chemical Name:
- Rifabutin
- Synonyms
- Ansamycin;Rifabutin-d7;RIFABUTINE;Rifabutin (LM427);lm427;RIFABUTIN;Mycobutin;Rifabatin;Levobutin;Ansatipine
- CBNumber:
- CB0702764
- Molecular Formula:
- C46H62N4O11
- Molecular Weight:
- 847
- MDL Number:
- MFCD00866816
- MOL File:
- 72559-06-9.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Rifabutin >98% (HPLC), powder | R3530 | 5mg | $124 |
| Rifabutin | PHR2611 | 100MG | $310 |
| Rifabutin | 1603800 | 50mg | $501 |
| Rifabutin European Pharmacopoeia (EP) Reference Standard | Y0000149 | 150 mg | $236 |
| Rifabutin >98.0%(HPLC) | R0211 | 100mg | $147 |
| More product size | |||
| Melting point | 169-171°C |
|---|---|
| Boiling point | 969.6±65.0 °C(Predicted) |
| Density | 1.33±0.1 g/cm3(Predicted) |
| storage temp. | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| solubility | DMSO: >5mg/mL |
| form | powder |
| pka | 3.31±0.70(Predicted) |
| color | Dark Red to Dark Purple |
| Water Solubility | 0.19g/L(temperature not stated) |
| BCS Class | 2 |
| Stability | Hygroscopic and Light Sensitive |
| InChIKey | ATEBXHFBFRCZMA-VXTBVIBXSA-N |
| CAS DataBase Reference | 72559-06-9 |
| FDA UNII | 1W306TDA6S |
| NCI Dictionary of Cancer Terms | ansamycin; Mycobutin; rifabutin |
| NCI Drug Dictionary | Mycobutin |
| ATC code | J04AB04 |
| UNSPSC Code | 12352200 |
| NACRES | NA.77 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302-H319 |
| Precautionary statements | P305+P351+P338 |
| PPE | Eyeshields, Gloves, type N95 (US) |
| WGK Germany | 3 |
| RTECS | VJ6700000 |
| HS Code | 2941906000 |
| Storage Class | 11 - Combustible Solids |
| Hazardous Substances Data | 72559-06-9(Hazardous Substances Data) |
Rifabutin price More Price(100)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | R3530 | Rifabutin >98% (HPLC), powder | 72559-06-9 | 5mg | $124 | 2026-04-30 | Buy |
| Sigma-Aldrich | PHR2611 | Rifabutin | 72559-06-9 | 100MG | $310 | 2026-04-30 | Buy |
| Sigma-Aldrich | 1603800 | Rifabutin | 72559-06-9 | 50mg | $501 | 2026-04-30 | Buy |
| Sigma-Aldrich | Y0000149 | Rifabutin European Pharmacopoeia (EP) Reference Standard | 72559-06-9 | 150 mg | $236 | 2025-07-31 | Buy |
| TCI Chemical | R0211 | Rifabutin >98.0%(HPLC) | 72559-06-9 | 100mg | $147 | 2026-04-30 | Buy |
Rifabutin Chemical Properties,Uses,Production
Description
Rifabutin, a rifamycin antibacterial derivative, is the first agent approved and introduced for the prevention of Mycobacterium avium complex (MAC) in AIDS patients. It is also indicated in combination chemotherapy for the prophylaxis and treatment of MAC infections in HIV positive patients and for newly diagnosed and chronic tuberculosis.
Chemical Properties
Red-Brown Powder
Originator
Archifar (Italy)
Uses
Semisynthetic derivative of Rifamycin S that inhibits nucleic acid synthesis. An antibacterial (tuberculostatic).
Uses
Antibiotic;RNA-polymerase inhibitor
Uses
Rifamycins are antibiotics that inhibit DNA-dependent RNA polymerases and are usually bactericidal against Gram-positive bacteria but bacteriostatic against Gram-negative bacteria. Rifamycins are also effective against Mycobacterium species, including M. tuberculosis. Rifabutin is a broad-spectrum rifamycin antibiotic that has applications against tuberculosis, H. pylori, M. avium complex, Chlamydia, and other bacteria. It is also useful in co-infections with human immunodeficiency virus, including tuberculosis.
Indications
Rifabutin (Mycobutin), an antibiotic related to rifampin, shares its mechanism of action, that is, inhibition of RNA polymerase. Rifabutin has significant activity in vitro and in vivo against M. avium-intracellular complex (MAC) isolates from both HIV-infected and non–HIV-infected individuals. It has better activity against MAC organisms than rifampin. Rifabutin is active against M. tuberculosis, including some rifampinresistant strains, such as M.leprae and M.fortuitum. It has a spectrum of activity against gram-positive and gramnegative organisms similar to that of rifampin. The molecular basis for resistance to rifabutin is shared by both rifampin and rifabutin; this explains the virtually complete cross-resistance that occurs between these drugs.
Definition
ChEBI: Rifabutin is a member of rifamycins. It has a role as an antitubercular agent.
brand name
Mycobutin (Pharmacia & Upjohn).
Antimicrobial activity
The activity is similar to that of rifampicin, but it is more active against the Mycobacterium avium complex (MIC 0.01–2 mg/L) and several other atypical mycobacteria. It inhibits the replication of human immunodeficiency virus 1 (HIV-1) in concentrations (10 mg/L) that are not toxic to lymphoid cells, but no efficacy on HIV infections has been demonstrated.
Acquired resistance
The frequency of spontaneously resistant mutants in several bacterial species, including M. tuberculosis, M. leprae, Staphylococcus aureus and Chlamydia trachomatis, is somewhat lower than with rifampicin.
Pharmaceutical Applications
Rifabutine; ansamycin. Molecular weight: 847.02.
A semisynthetic spiropiperidyl derivative of rifamycin S, available for oral administration. It is slightly soluble in water and soluble in organic solvents.
Biochem/physiol Actions
Rifabutin is an antibiotic; antitumor. Rifabutin interferes with HSP-90 molecular chaperone, enhances ubiquitination and protein degradation, and inactivates bacterial RNA polymerase.
Pharmacology
Rifabutin is well absorbed orally, and peak plasma
concentrations are reached in 2 to 3 hours. Because of
its lipophilicity, rifabutin achieves a 5- to 10-fold higher
concentration in tissues than in plasma. The drug has a
half-life range of 16 to 96 hours and is eliminated in
urine and bile.
Rifabutin appears as effective as rifampin in the
treatment of drug-susceptible tuberculosis and is used
in the treatment of latent tuberculosis infection either
alone or in combination with pyrazinamide. Clinical use
of rifabutin has increased in recent years, especially in
the treatment of HIV infection. It is a less potent
inducer of cytochrome 450 enzymes pathways than rifampin
and results in less drug interaction with the
protease inhibitors and nonnucleoside reverse transcriptase
inhibitors. Rifabutin is therefore commonly
substituted for rifampin in the treatment of tuberculosis
in HIV-infected patients. Another important use of rifabutin
in the HIV-infected population is prevention
and treatment of disseminated MAC.
Pharmacokinetics
Oral absorption:12–20%
Cmax 300 mg oral :0.38 mg/L after 3.3 h
Plasma half-life:16 h
Volume of distribution:9.3 L/kg
Plasma protein binding: 85%
absorption and distribution
Oral absorption is rapid but incomplete, with considerable interpatient variation. It is well distributed, concentrations in many organs being higher than that in plasma. The average concentration in lungs is 6.5 times the simultaneous plasma concentration.
Metabolism and excretion
Rifabutin is mainly metabolized to the active desacetyl derivative, although several other oxidation products have been detected in urine, where some 10% of the dose is eliminated. About 30–50% of the dose can be recovered from the feces. Elimination from plasma is biphasic, with a terminal half-life of 45 h. The drug is a weak inducer of hepatic enzymes. The rate of metabolism increases, and the plasma area under the concentration–time curve (AUC) declines as the treatment continues.
Clinical Use
Rifabutin, the spiroimidazopiperidyl derivative of rifamycin B was approved in the United States for the prophylaxis of disseminated MAC in AIDS patients on the strength of clinical trials establishing its effectiveness. The activity of rifabutin against MAC organisms greatly exceeds that of rifamycin. This rifamycin derivative is not effective, however, as monotherapy for existing disseminated MAC disease. Rifabutin is a very lipophilic compound with a high affinity for tissues. Its elimination is distribution limited, with a half-life averaging 45 hours (range, 16 69 hours). Approximately 50% of an orally administered dose of rifabutin is absorbed, but the absolute oral bioavailability is only about 20%. Extensive first-pass metabolism and significant biliary excretion of the drug occur, with about 35% and 53% of the orally administered dose excreted, largely as metabolites, in the feces and urine, respectively. Despite its greater potency against M. tuberculosis in vitro, rifabutin is considered inferior to rifampin for the short-term therapy of tuberculosis because of its significantly lower plasma concentrations.
Although rifabutin is believed to cause less hepatotoxicityand induction of cytochrome P450 enzymes than rifampin,these properties should be borne in mind when the drug is usedprophylactically. Rifabutin and its metabolites are highly coloredcompounds that can discolor skin, urine, tears, feces, etc.
Clinical Use
Prevention of infections with M. avium complex in AIDS patients
Treatment of non-tuberculous mycobacterial disease (in combination with other agents)
Rifabutin in combination with other agents has been proposed as a rescue therapy after Helicobacter pylori treatment failures.Although some efficacy has been observed in the treatment of tuberculosis, its use for this condition is not recommended.
Side effects
The adverse effects that most frequently result in discontinuation of rifabutin include GI intolerance, rash, and neutropenia. Rifabutin levels will be increased with concurrent administration of fluconazole and clarithromycin, resulting in anterior uveitis, polymyalgia syndrome, and a yellowish-tan discoloration of the skin (pseudojaundice). Other adverse reactions are similar to those of rifampin, such as hepatitis, red-orange discoloration of body fluids, and drug interactions due to effects on the hepatic P450 cytochrome enzyme system.
Side effects
Rash (4% of patients), gastrointestinal intolerance (3%) and neutropenia (2%) are fairly common and may require discontinuation of treatment. Uveitis and general arthralgia are rare with a 300 mg dosage, but frequent with higher dosages, especially with concomitant use of fluconazole or macrolide antibiotics.
Drug interactions
Potentially hazardous interactions with other drugs
Anti-arrhythmics: metabolism of disopyramide,
and propafenone accelerated; concentration of
dronedarone reduced.
Antibacterials: increased risk of side effects with
azithromycin; clarithromycin and other macrolides
increase concentration of rifabutin, resulting
in increased risk of uveitis - reduce rifabutin
dose; reduced concentration of dapsone and
clarithromycin.
Anticoagulants: reduced anticoagulant effect of
coumarins.
Antidiabetics: reduced antidiabetic effect of
tolbutamide; possibly reduced antidiabetic effect
with sulphonylureas.
Antiepileptics: reduced concentration of
fosphenytoin, phenytoin and carbamazepine.
Antifungals: fluconazole, triazoles, posaconazole and
voriconazole increase the concentration of rifabutin
resulting in increased risk of uveitis - reduce
rifabutin dose; rifabutin reduces concentration
of posaconazole, voriconazole and itraconazole -
increase voriconazole dose, avoid with isavuconazole
and itraconazole.
Antipsychotics: possibly reduced aripiprazole
concentration - increase dose of aripiprazole.
Antivirals: atazanavir darunavir, fosamprenavir,
saquinavir and tipranavir and possibly nevirapine
increase concentration of rifabutin - halve or
reduce dose of rifabutin; efavirenz reduces the
concentration of rifabutin - increase dose of
rifabutin; concentration of both drugs reduced
with etravirine; indinavir increases rifabutin
concentration - avoid; concentration of indinavir
reduced - increase indinavir dose; concentration of
elvitegravir reduced and active metabolite of rifabutin
increased - reduce dose of rifabutin; concentration
of rilpivirine reduced - increase rilpivirine dose to 50mg once daily; ritonavir increases the concentration
of rifabutin resulting in increased risk of uveitis -
reduce rifabutin dose; concentration of saquinavir
reduced and concentration of rifabutin increased -
reduce rifabutin dose; concentration of daclatasvir
and simeprevir possibly reduced - avoid; avoid with
ledipasvir, sofosbuvir and telaprevir.
Atovaquone: concentration of atovaquone reduced
(possible therapeutic failure of atovaquone).
Ciclosporin: possibly reduced ciclosporin levels.
Cobicistat: concentration of cobicistat reduced -
adjust cobicistat dose.
Corticosteroids: reduced level of corticosteroids -
double steroid dose. Give as twice daily dosage.
Cytotoxics: possibly reduced concentration
of axitinib (increase axitinib dose), bosutinib,
cabazitaxel, ceritinib, crizotinib, lapatinib, olaparib,
panobinostat, ponatinib and vemurafenib - avoid.
Guanfacine: concentration of guanfacine possibly
reduced - increase dose of guanfacine.
Hormone antagonists: concentration of abiraterone
possibly reduced - avoid.
Ivacaftor: concentration of ivacaftor possibly reduced
- avoid.
Oestrogens and progestogens: reduced contraceptive
effect due to increased metabolism.
Sirolimus: reduced sirolimus concentration - avoid.
Tacrolimus: possibly reduced tacrolimus trough
concentration.
Ulipristal: possibly reduced contraceptive effect -
avoid.
Metabolism
Rifabutin is rapidly metabolised in the liver by the
cytochrome P450 isoenzyme CYP3A4 mainly to active
25-O-deacetyl and 31-hydroxy metabolites. Rifabutin
induces its own metabolism resulting in a lower AUC
after 4 weeks of continuous treatment than after the first
few doses.
About 53% of a dose is found in the urine, mainly as
metabolites and about 30% of a dose is excreted in the
faeces.
Rifabutin Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Cangzhou Kangrui Pharma Tech Co. Ltd., | +86-18632776803 +86-13833998158 | cangzhoukangrui@126.com | China | 773 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615350571055 | Sibel@chuanghaibio.com | China | 8738 | 58 |
| Sinoway Industrial co., ltd. | +86-0592-5800732 +86-13806035118 | xie@china-sinoway.com | China | 1369 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-2158073036 | info@dakenam.com | China | 13901 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21587 | 55 |
| Nanjing ChemLin Chemical Industry Co., Ltd. | 025-83697070 | product@chemlin.com.cn | CHINA | 3009 | 60 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29815 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19936 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49979 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354; +17819995354 | marketing@targetmol.com | United States | 32466 | 58 |
Related articles
- Side-effects of rifabutin
- Similar to rifampicin, rifabutin (also known as ansamycin LM 427) is a derivative of rifamycin S. The most important property ....
- Mar 28,2022
View Lastest Price from Rifabutin manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-06-06 | Rifabutin
72559-06-9
|
0.99 | RongNa Biotechnology Co.,Ltd | ||||
 |
2026-06-05 | Rifabutin
72559-06-9
|
1g | 95%-102% | 100kgs | WUHAN FORTUNA CHEMICAL CO., LTD | ||
 |
2026-06-03 | Rifabutin
72559-06-9
|
1kg | 98% | 20TONS | Sinoway Industrial co., ltd. |